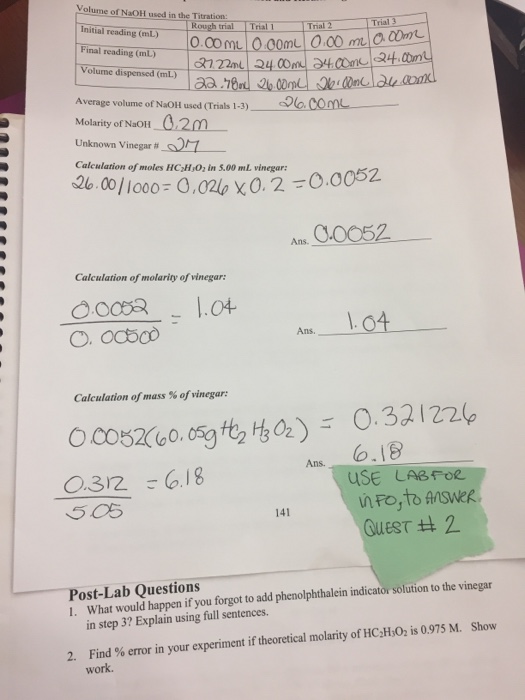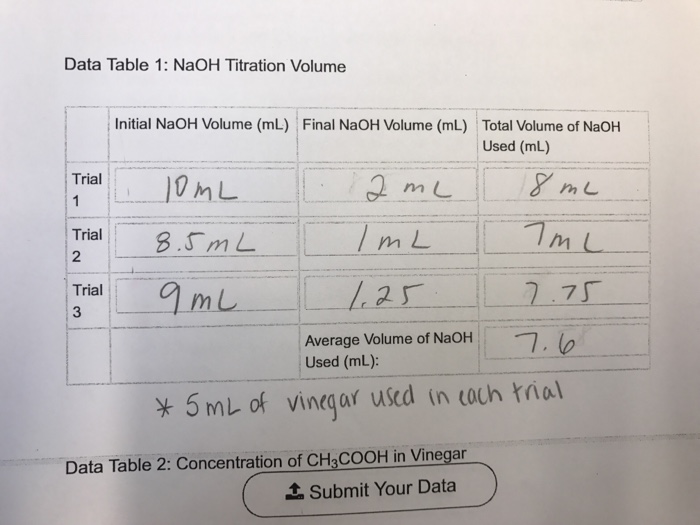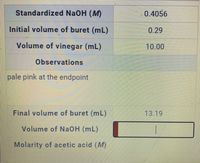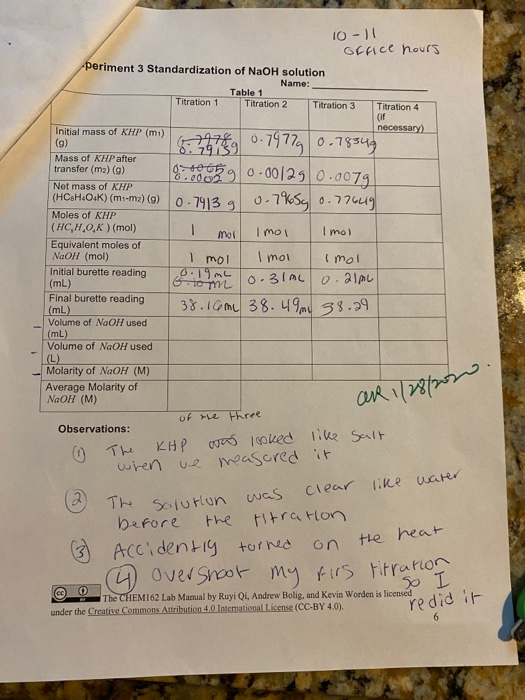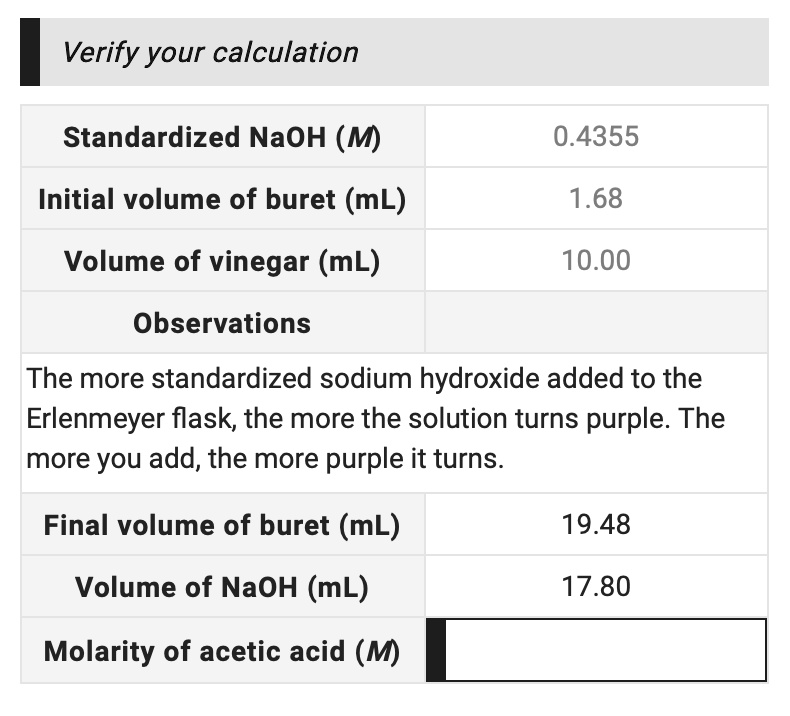
SOLVED: Verify your calculation Standardized NaOH (M) 0.4355 Initial volume of buret (mL) 1.68 Volume of vinegar (mL) 10.00 Observations The more standardized sodium hydroxide added to the Erlenmeyer flask; the more
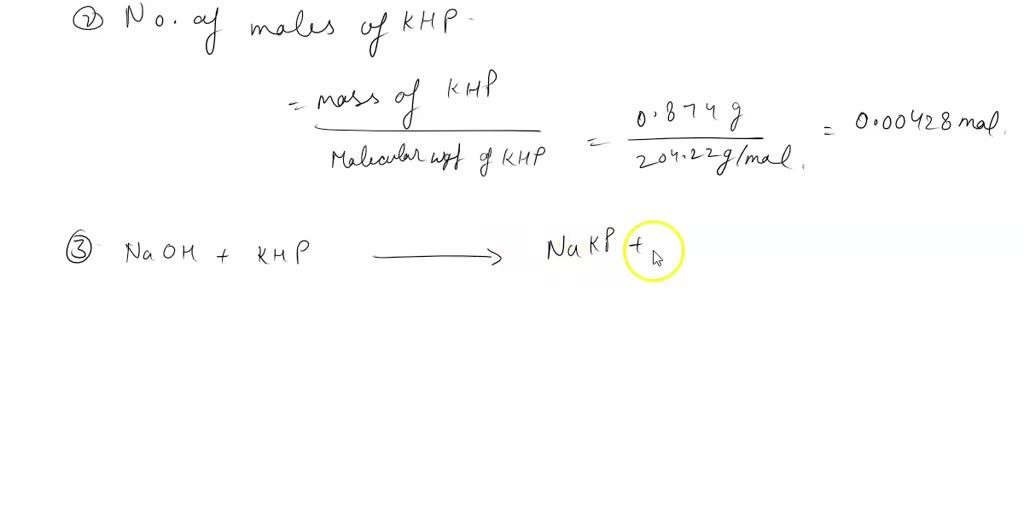
SOLVED: Determine the volume of NaOH used by subtracting the final burette volume from the initial burette volume. Calculate the number of moles of KHP in your sample from the mass of

How can i calculate the volume of NaOH in this question? I know the pH has to be equal to the pKa, because is a buffer, so pH= 4.74. But for the

What volume of 0.2 M NaOH (in ml) solution should be mixed to 500 ml of 0.5 M NaOH solution - YouTube

The titration data plotted as conductivity vs. volume of NaOH for the... | Download Scientific Diagram
Partial molar volume of NaOH(aq) at infinite dilution at saturation... | Download Scientific Diagram

Draw the pH vs volume NaOH for titrating HOOCCH_2CH_2OH with NaOH. What is the pH at the endpoint? What will be different when HOOCCH_2COOH is titrated with NaOH? | Homework.Study.com

Determine the volume of 0.125 M NaOH required to titrate to the equivalence point 25.0 mL of a - YouTube

Question Video: Calculating the Volume of Sulfuric Acid That Completely Neutralizes a Given Volume and Concentration of Sodium Hydroxide | Nagwa

The variation in pH with volume of NaOH added when an wear with volume of NaOH added when an weak acid HA is titrated with OL M NaOH represented as follows