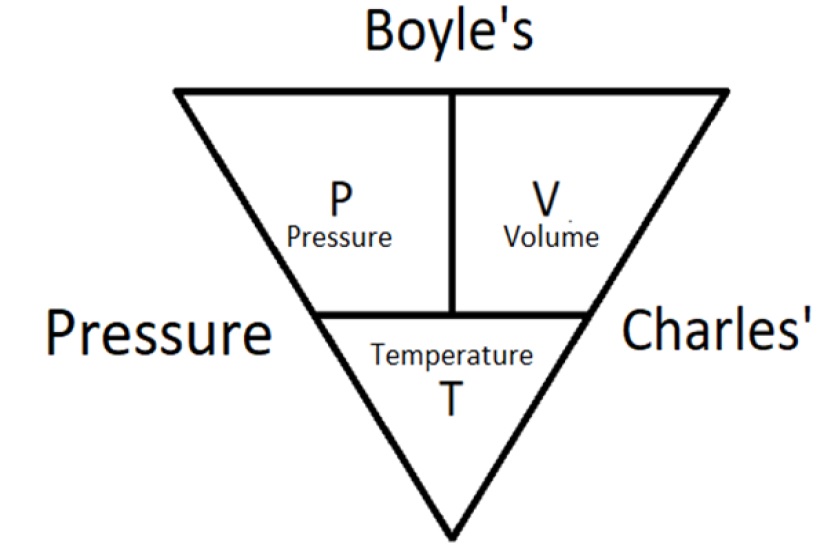
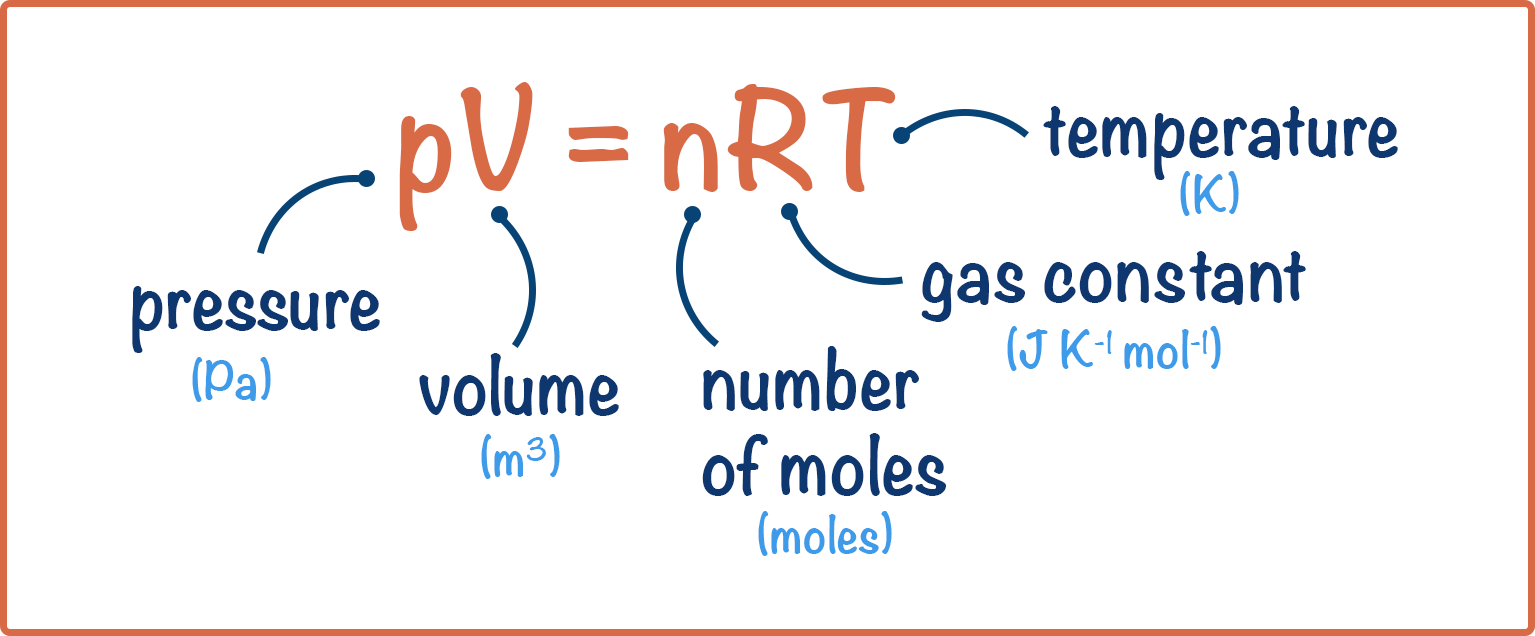
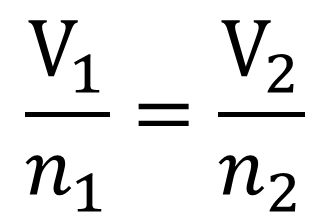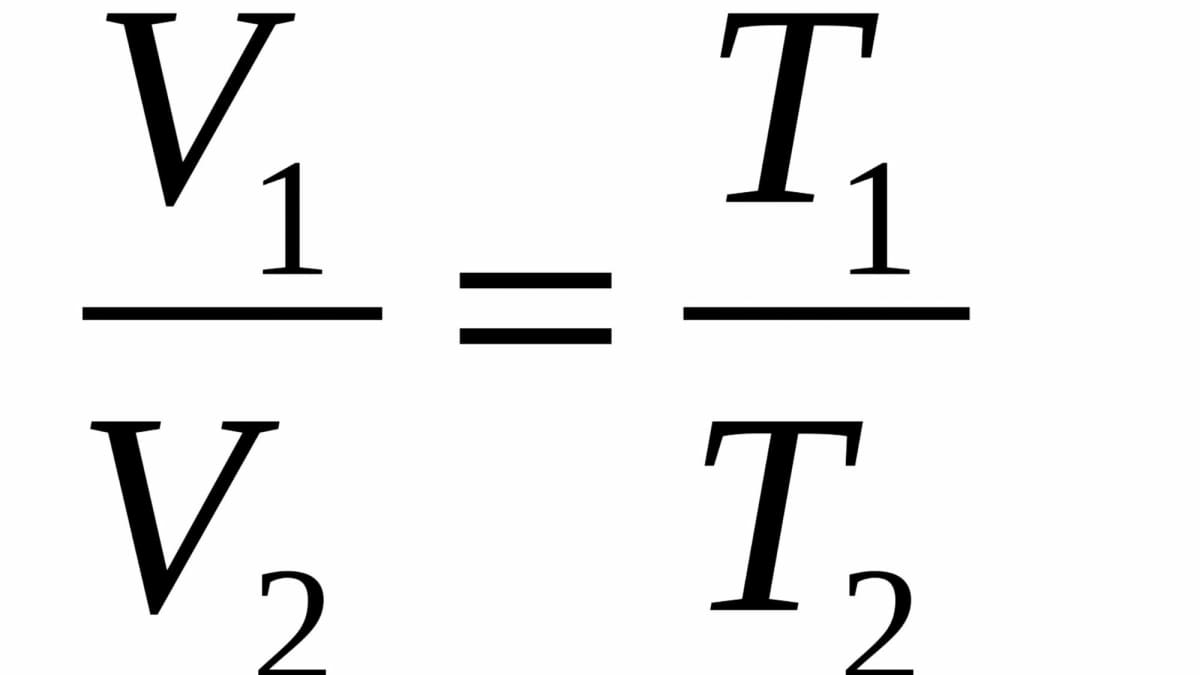A container with a volume of 18 L contains a gas with a temperature of 220^o C. If the temperature of the gas changes to 320 ^o K without any change in

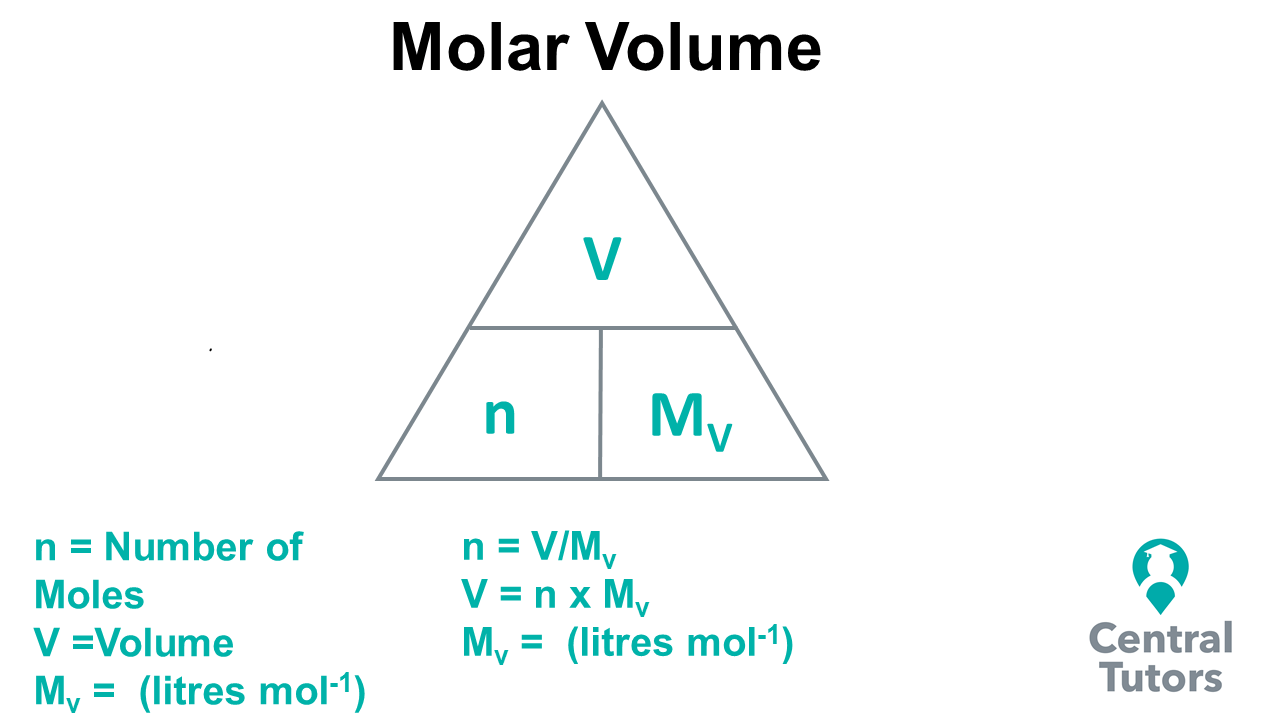
molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises
Formula ideale per la legge del gas. Pressione, volume, quantità di sostanza, costante ideale del gas e temperatura. Risorse di fisica per insegnanti e studenti Immagine e Vettoriale - Alamy