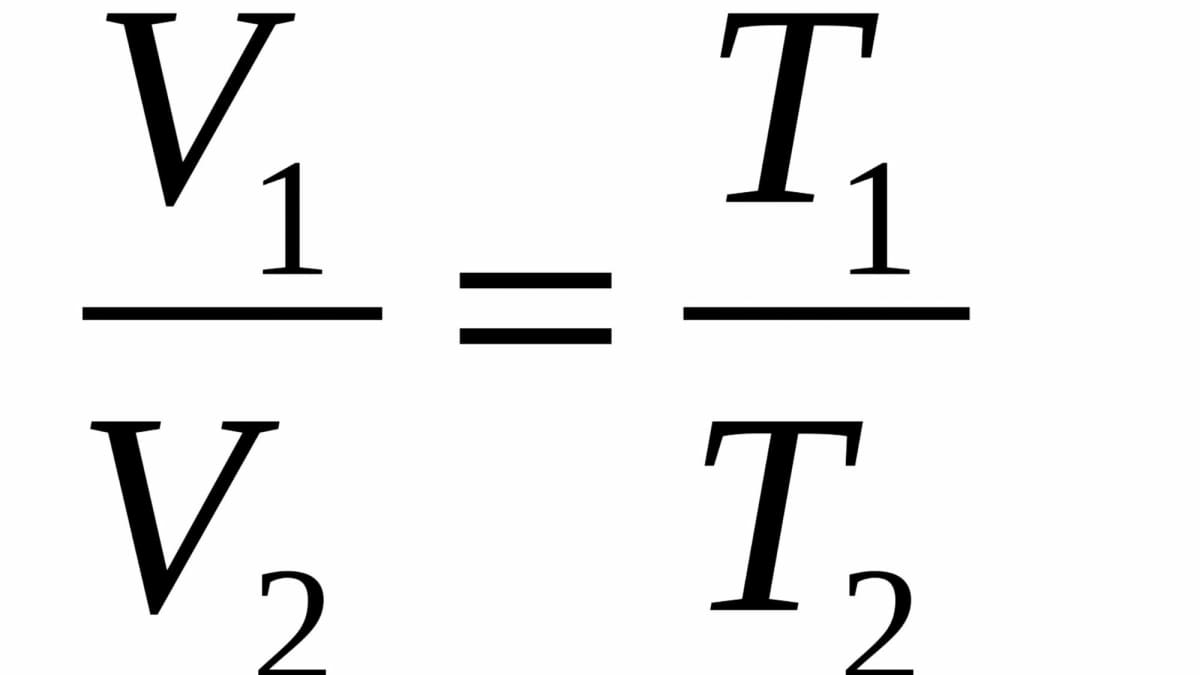Title: Lesson 9: Relationship between Volume, Temperature and Pressure Learning Objectives: Recall the different gas laws Identify when to use the different. - ppt download
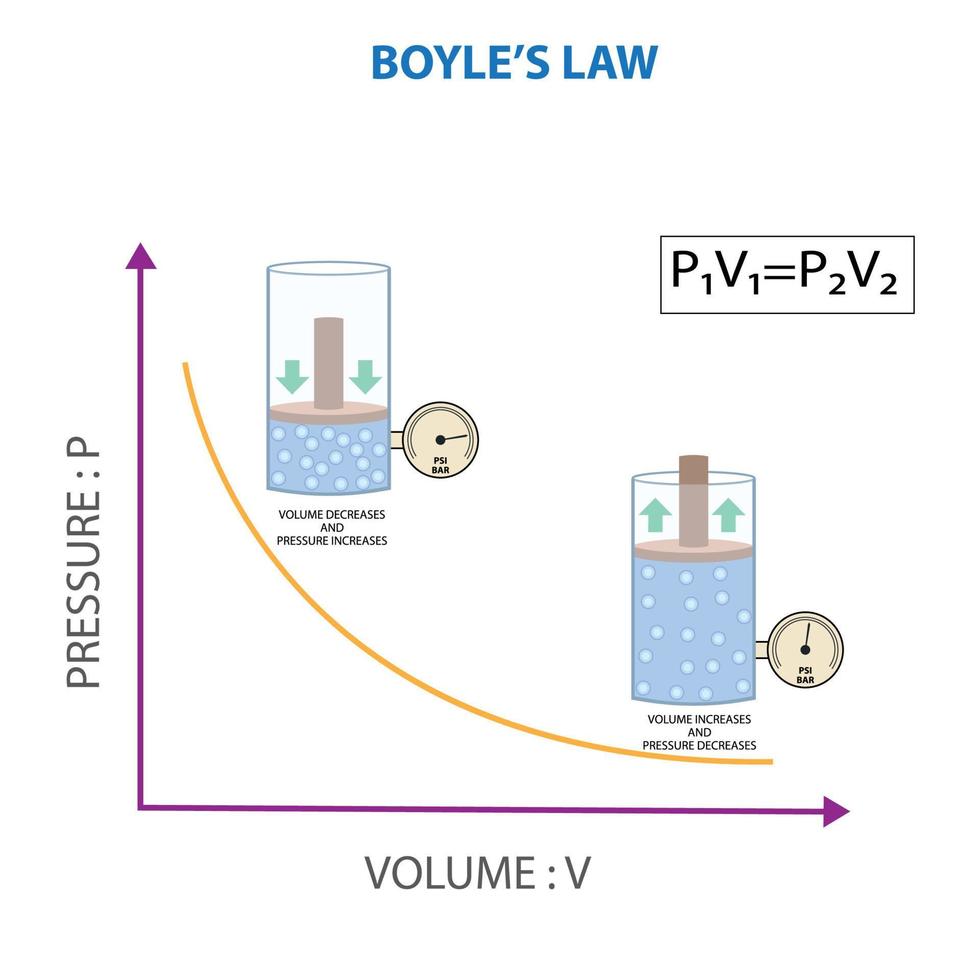
Boyle's Law, Relationship between pressure and volume of gas at constant temperature 21669324 Vector Art at Vecteezy
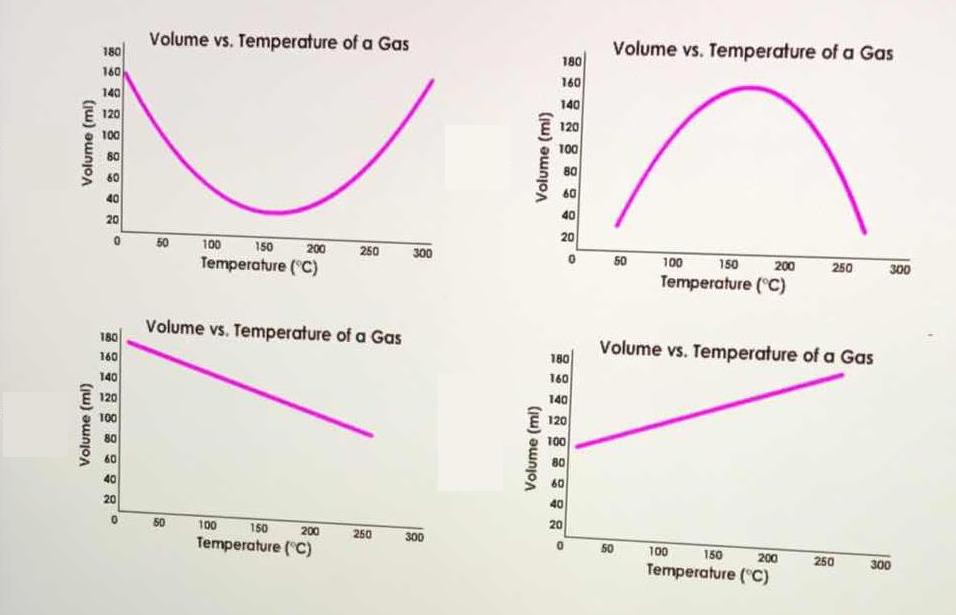
Which graph shows the relationship between the temperature and volume of a gas according to Charles's law? | Socratic

SOLVED: Derive the volume-temperature relationship according to V. with Cp/Cv = y,for the reversible adiabatic expansion of an ideal gas from the pressure-volume relationship P1VY =P2VX

Which graph represents the relationship between pressure and volume for an ideal gas at constant - Brainly.in
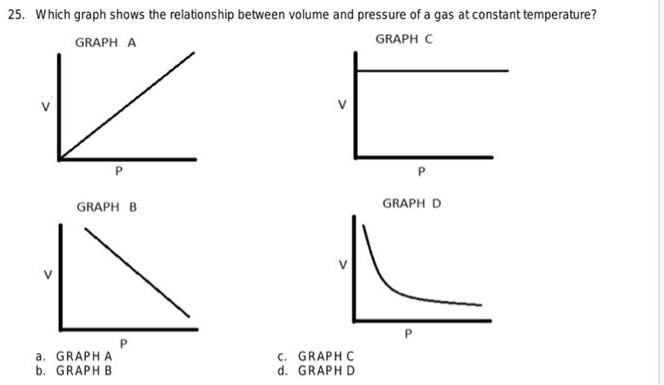
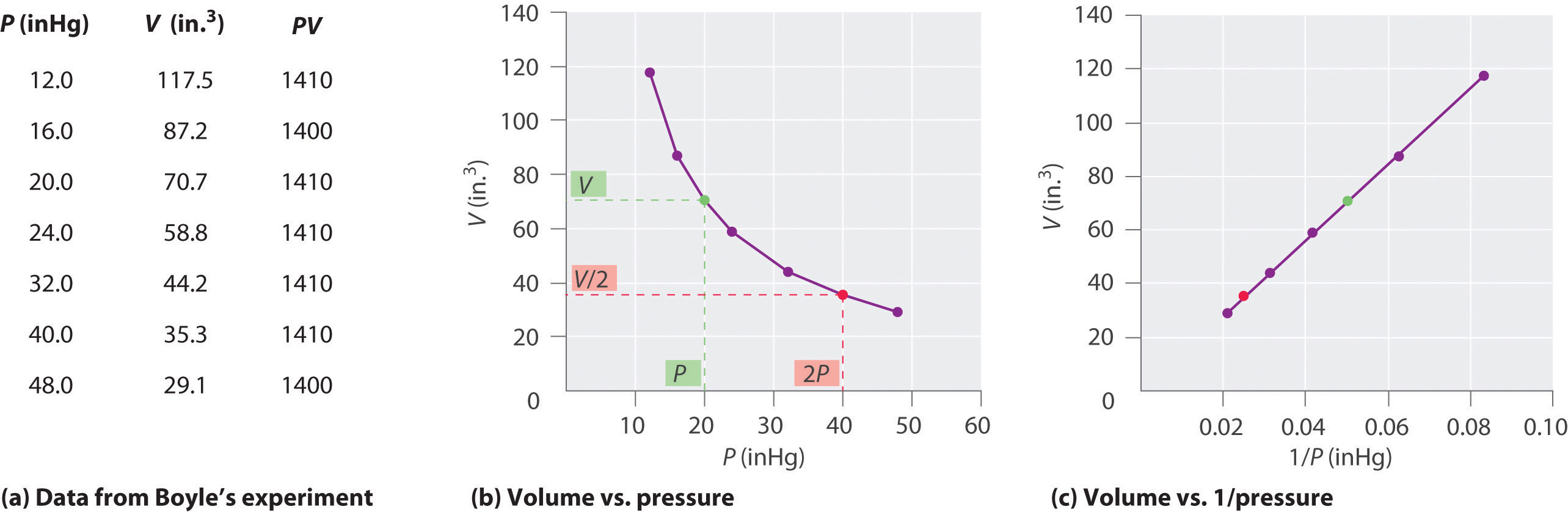
SOLVED: Which graph shows the relationship between volume and pressure of a gas at constant temperature? GRAPH GRAPH GRAPH GRAPH GRAPH GRAPH B GRAPH € GRAPH D

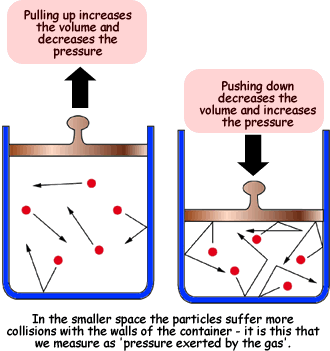
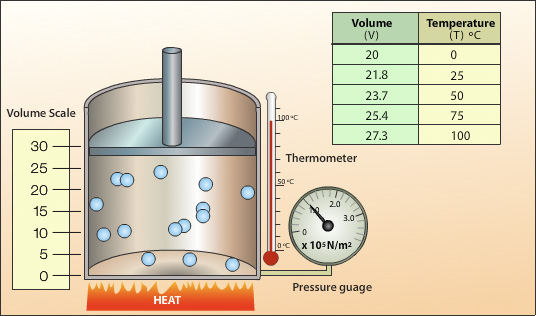


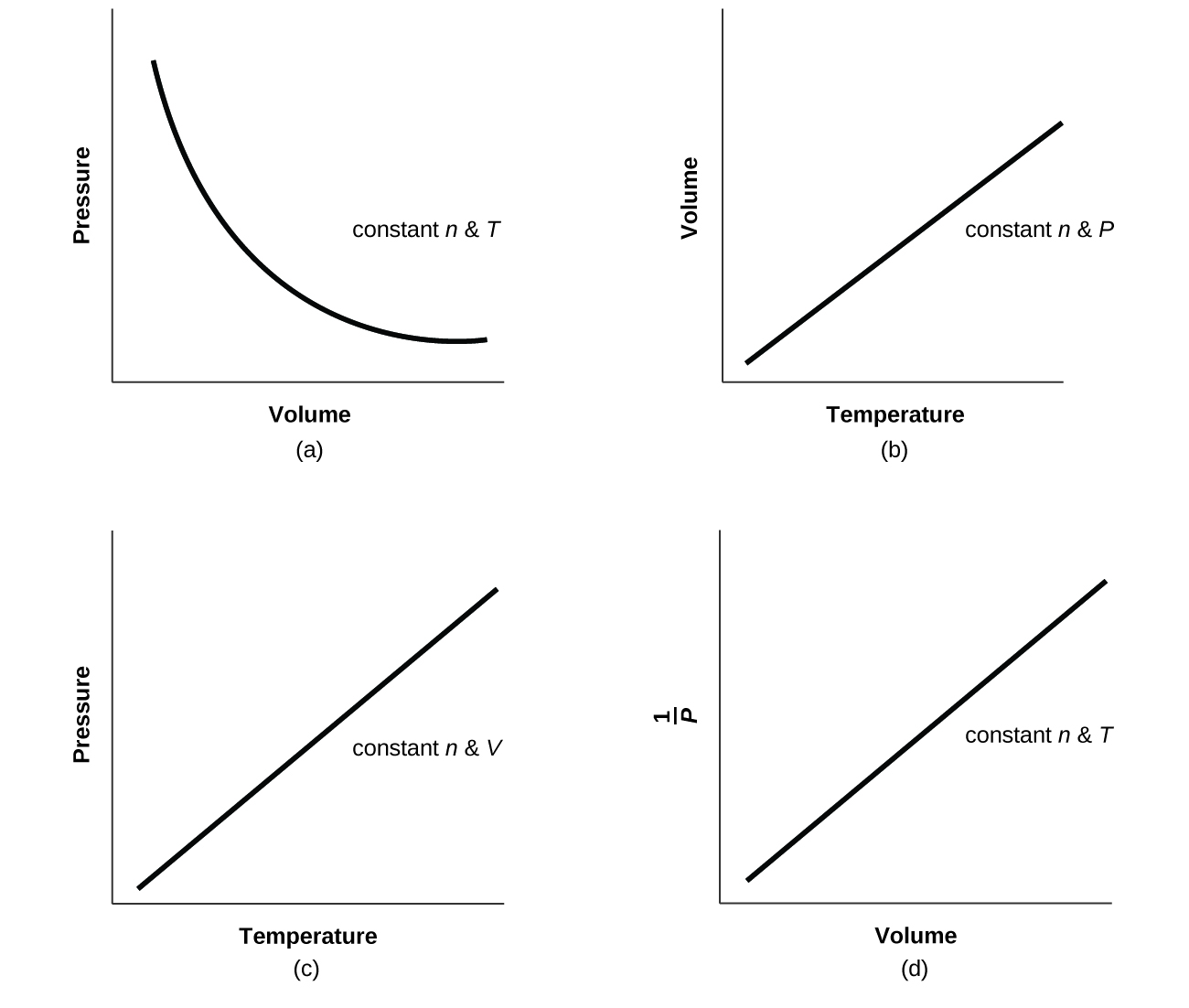
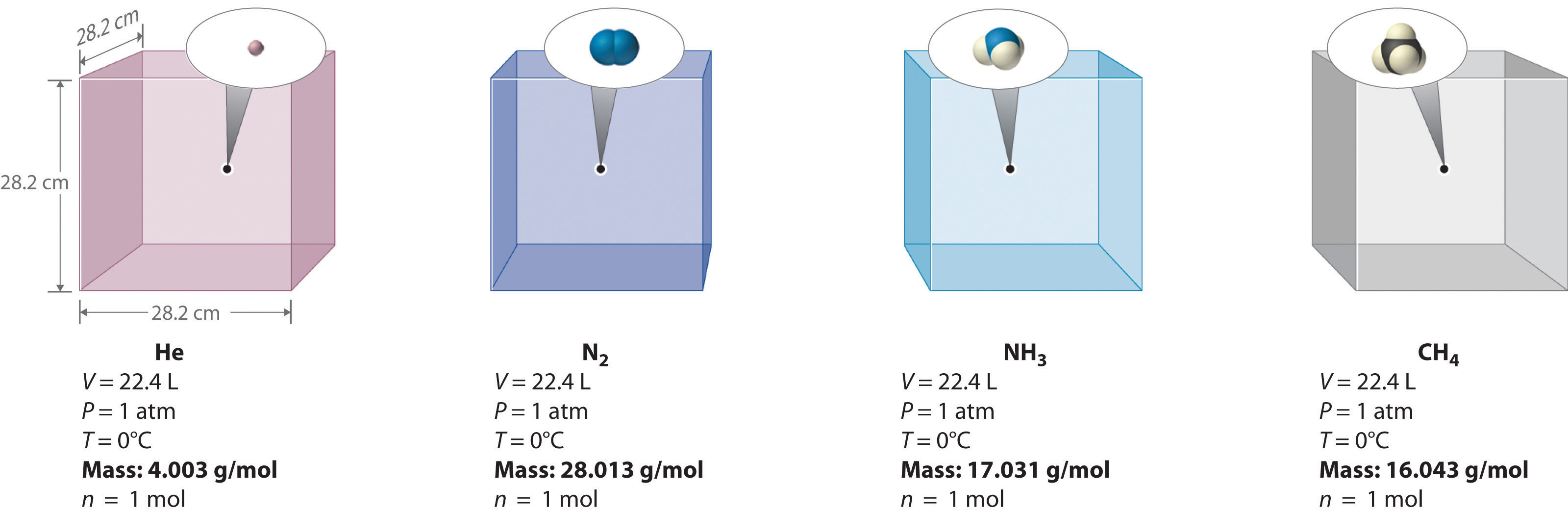




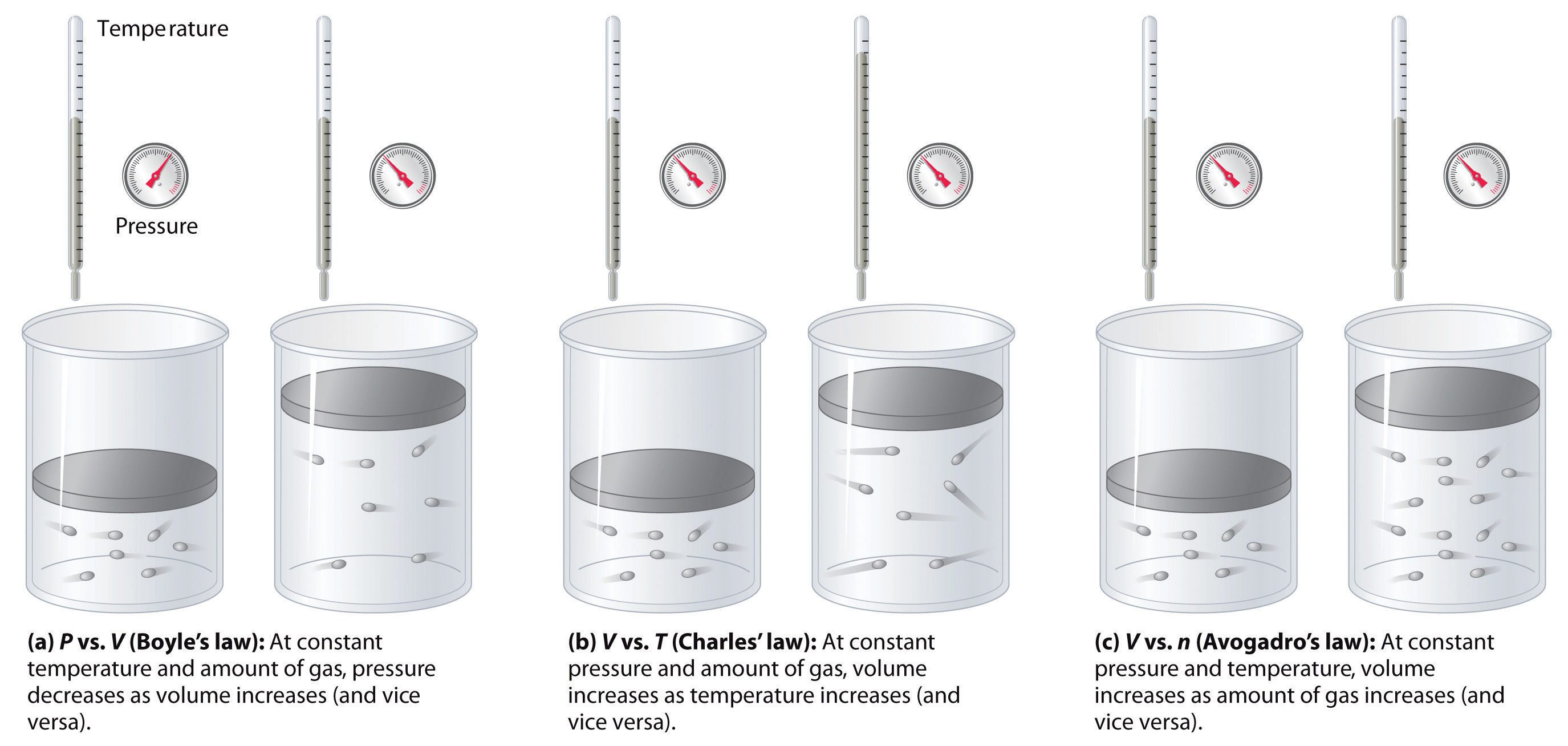

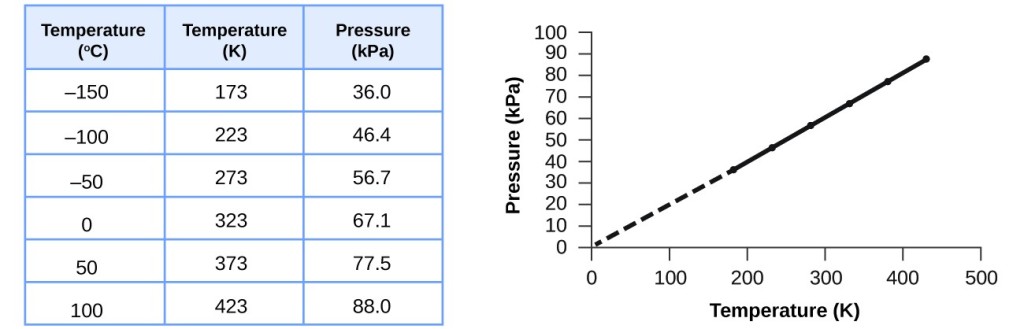
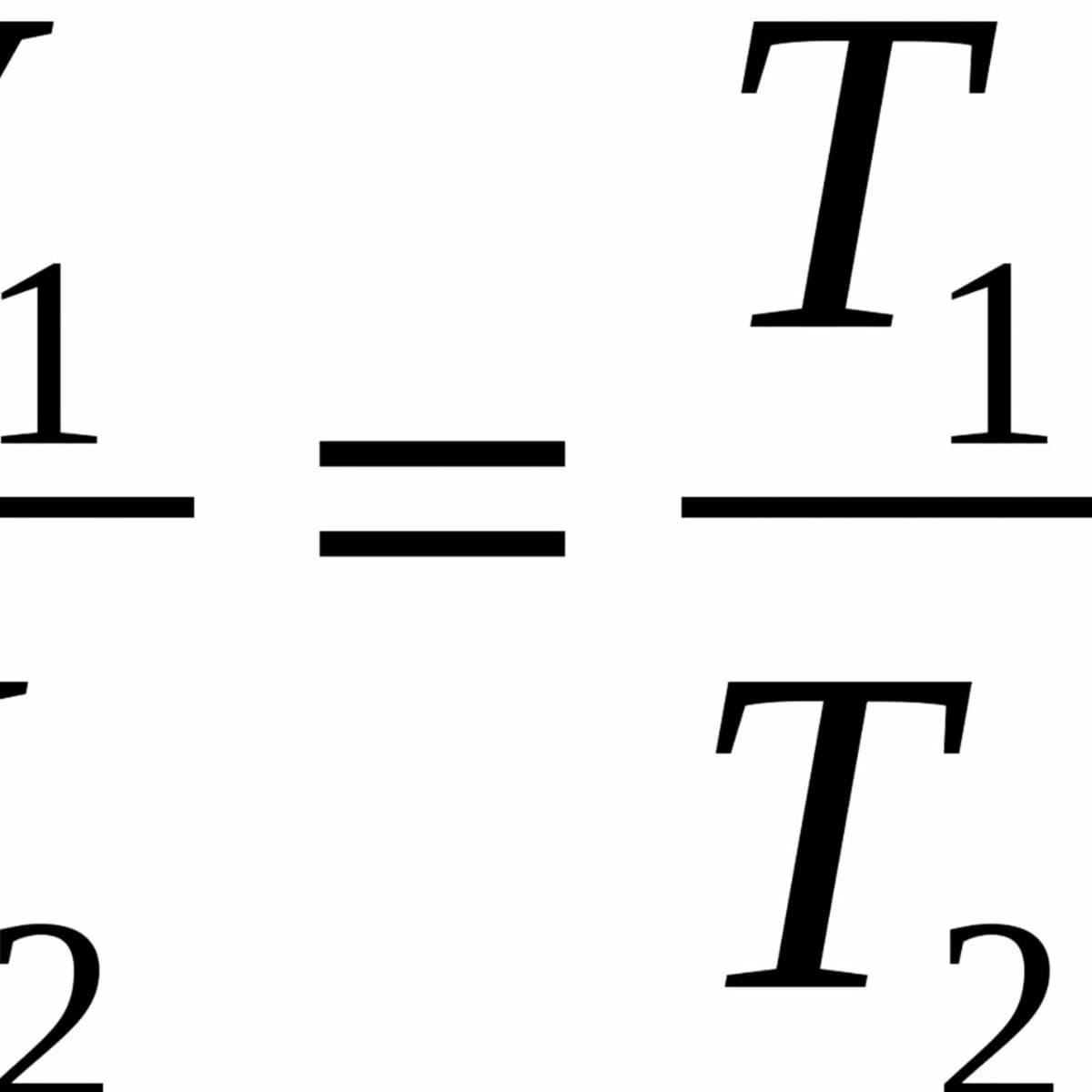
.jpg?revision=1)