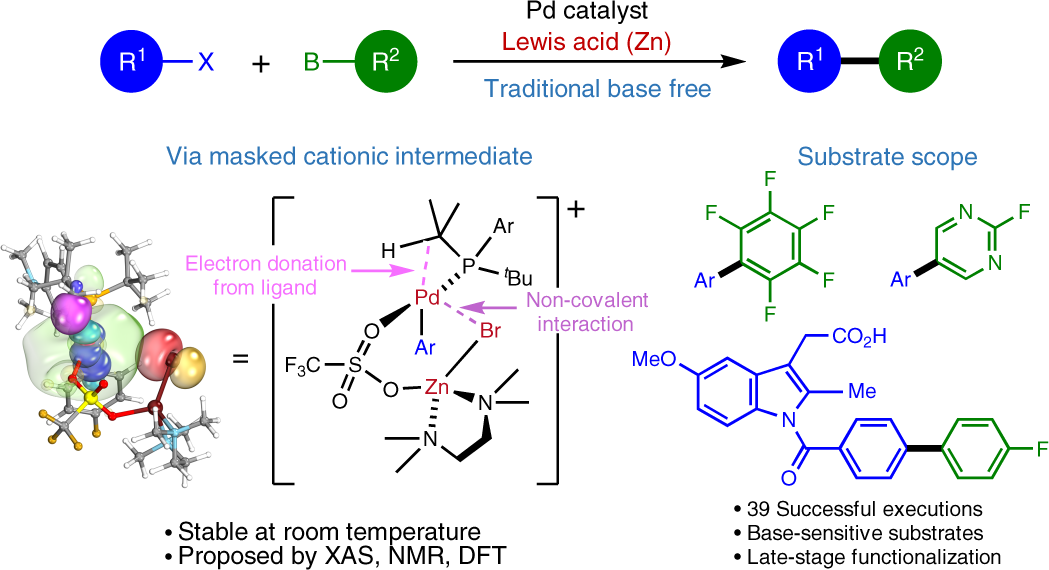
Molecules | Free Full-Text | Efficient Suzuki–Miyaura C-C Cross-Couplings Induced by Novel Heterodinuclear Pd-bpydc-Ln Scaffolds

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect
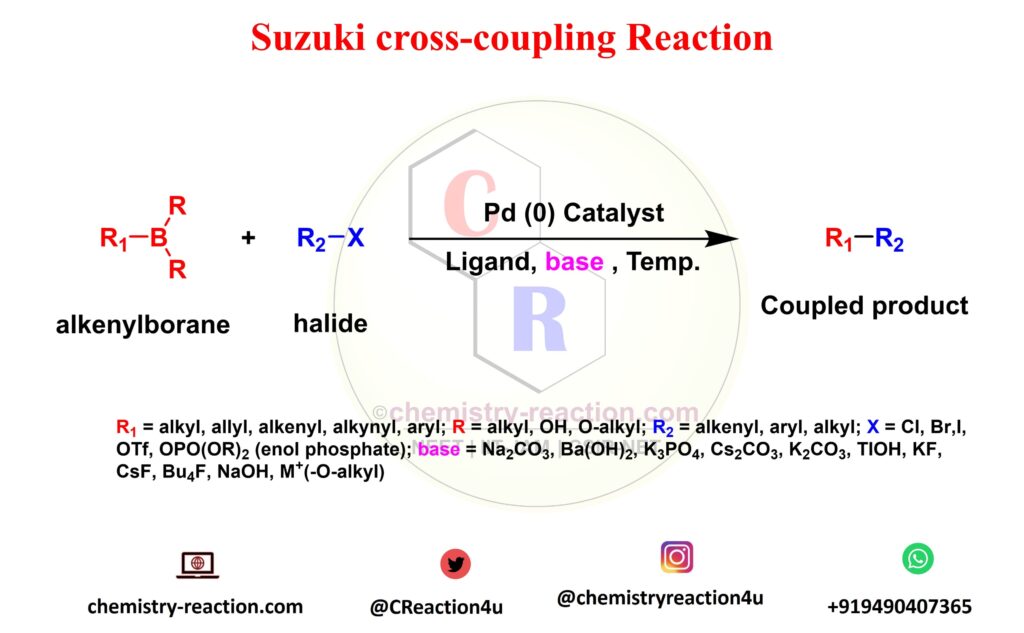
Suzuki–Miyaura cross coupling reaction: recent advancements in catalysis and organic synthesis - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/D0CY02059A
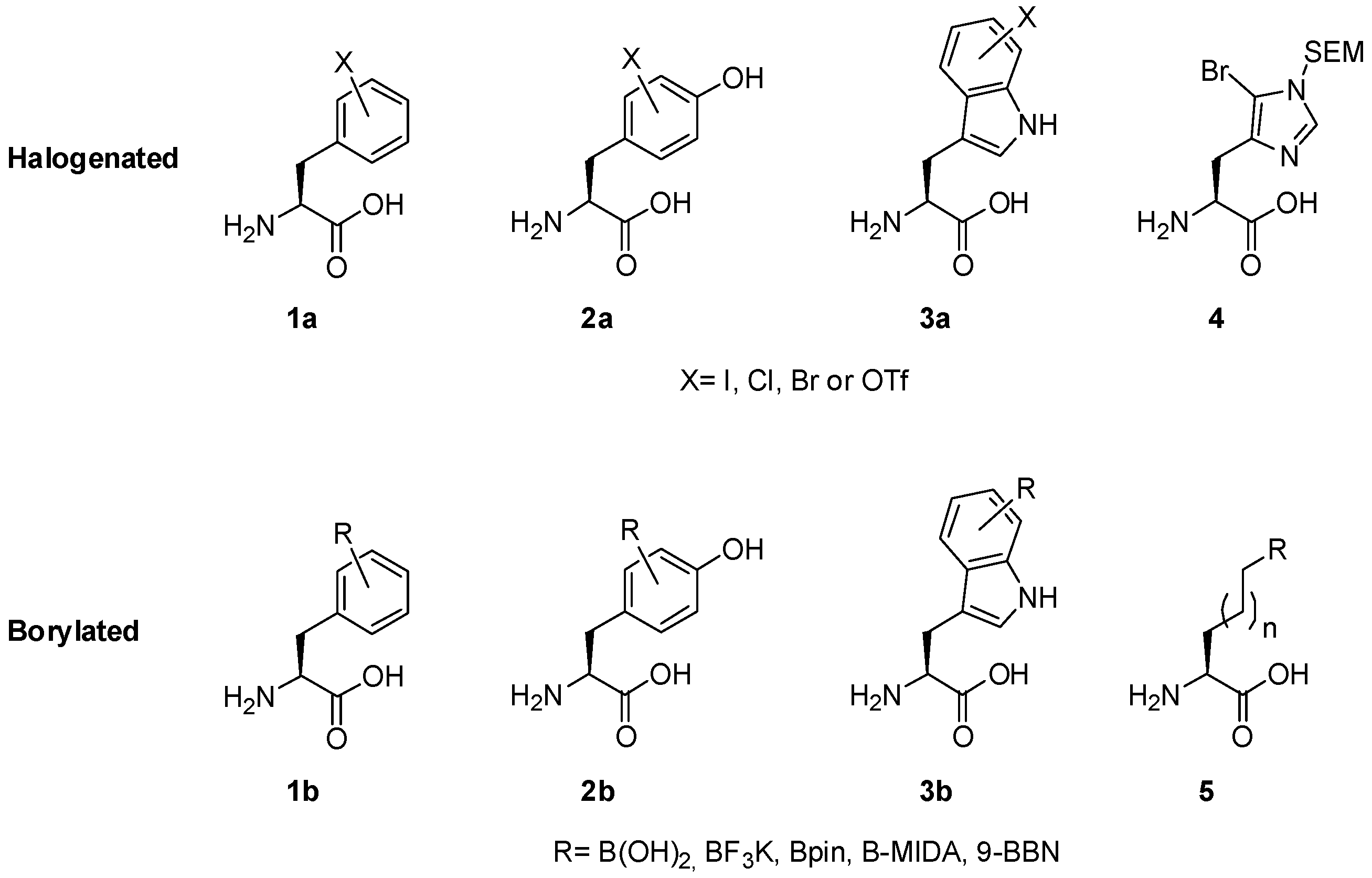
Catalysts | Free Full-Text | The Suzuki–Miyaura Cross-Coupling as a Versatile Tool for Peptide Diversification and Cyclization

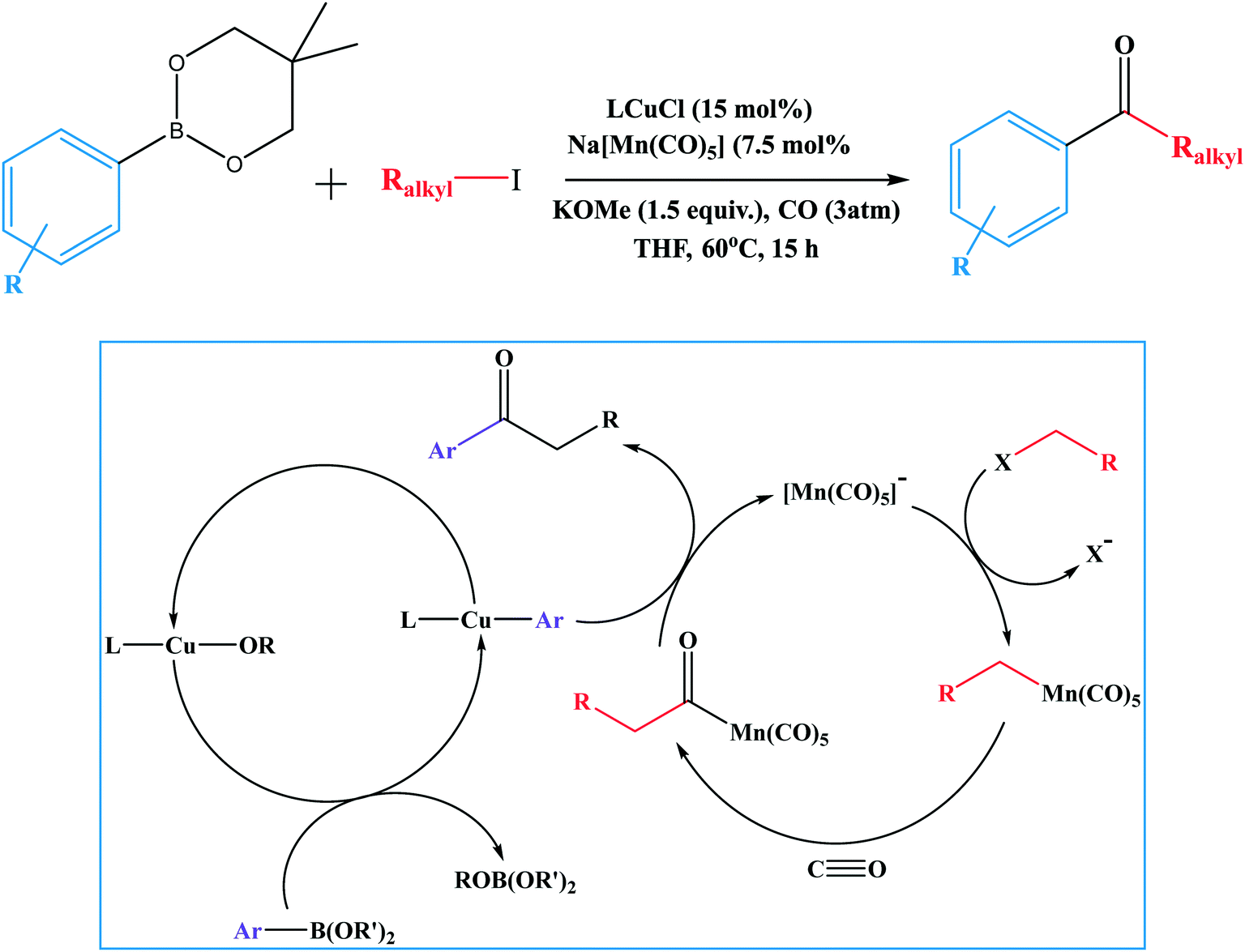
Suzuki–Miyaura cross-couplings for alkyl boron reagent: recent developments—a review | Future Journal of Pharmaceutical Sciences | Full Text
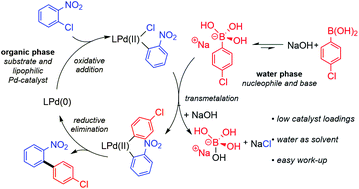
Pd-Catalysed Suzuki–Miyaura cross-coupling of aryl chlorides at low catalyst loadings in water for the synthesis of industrially important fungicides - Green Chemistry (RSC Publishing)

Unsymmetrical Biheteroaryls via Suzuki–Miyaura Cross-Couplings - Thieme Chemistry - Georg Thieme Verlag KG
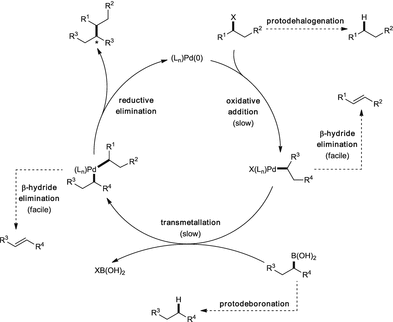
At the Forefront of the Suzuki–Miyaura Reaction: Advances in Stereoselective Cross-Couplings | SpringerLink
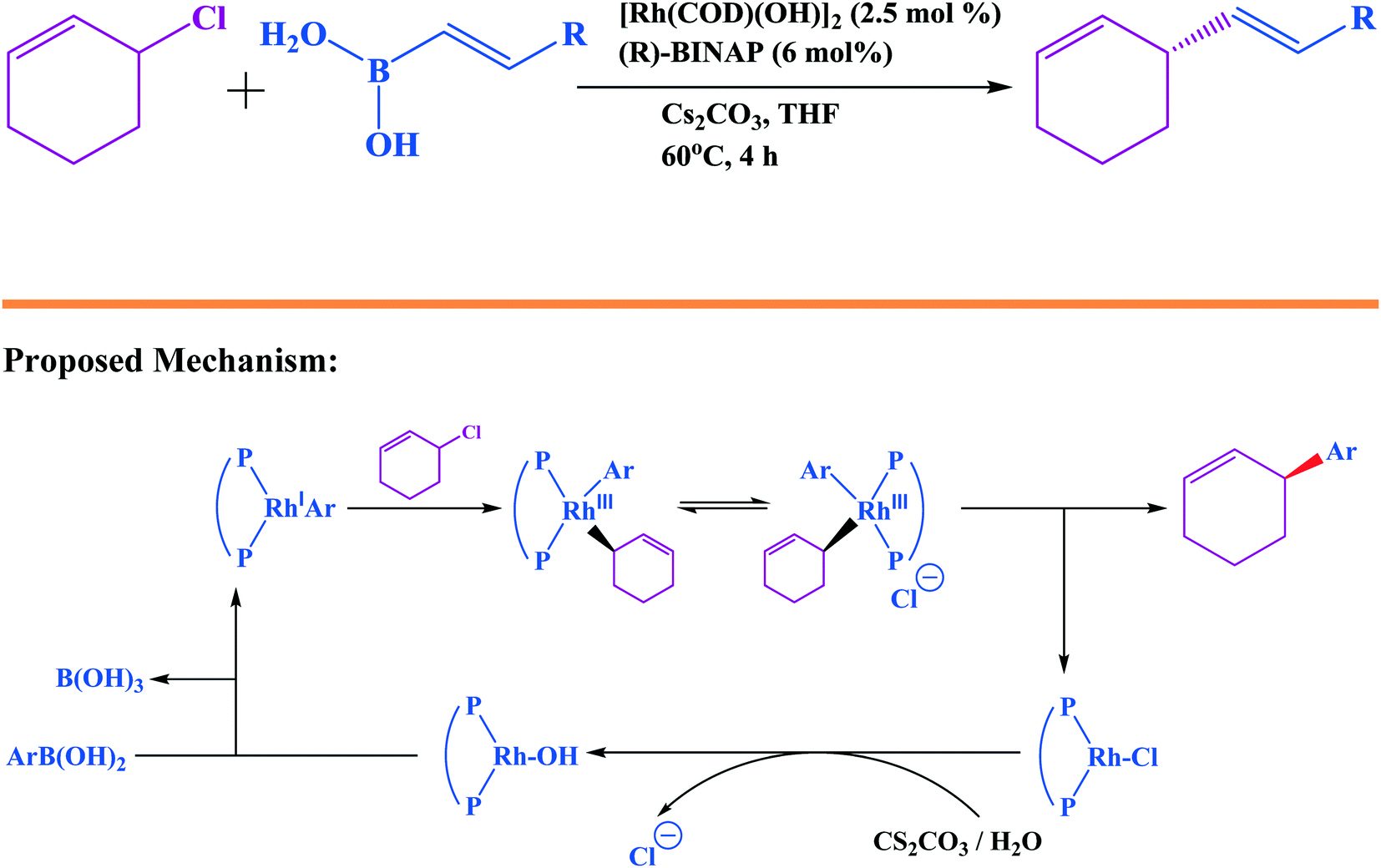
Suzuki–Miyaura cross coupling reaction: recent advancements in catalysis and organic synthesis - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/D0CY02059A

Suzuki–Miyaura cross-couplings for alkyl boron reagent: recent developments—a review | Future Journal of Pharmaceutical Sciences | Full Text
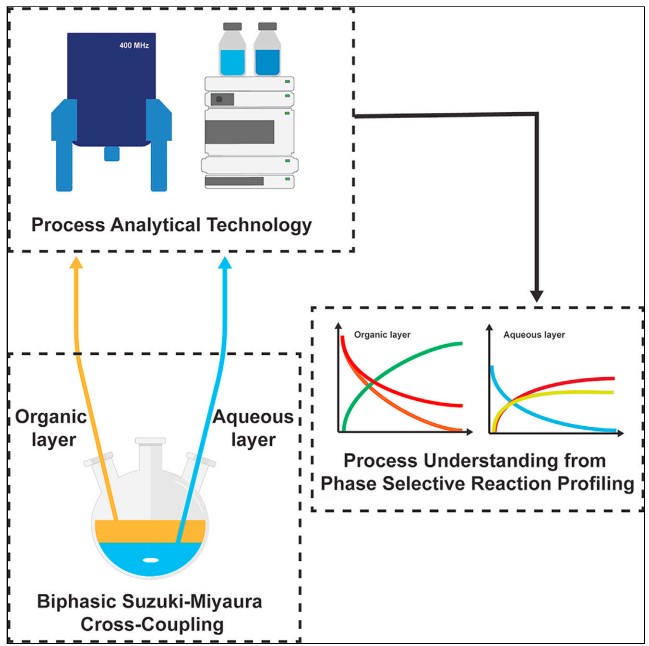
Liquid/liquid heterogeneous reaction monitoring: Insights into biphasic Suzuki-Miyaura cross-coupling - Vapourtec

Nickel-Catalyzed Suzuki–Miyaura Cross-Coupling in a Green Alcohol Solvent for an Undergraduate Organic Chemistry Laboratory | Semantic Scholar