![PDF] PHOTOCHEMICAL REDUCTION OF SILVER NITRATE TO NANO SILVER USING STANNOUS CHLORIDE, CTAB AND DAYLIGHT IRRADIATION | Semantic Scholar PDF] PHOTOCHEMICAL REDUCTION OF SILVER NITRATE TO NANO SILVER USING STANNOUS CHLORIDE, CTAB AND DAYLIGHT IRRADIATION | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c2436b224b00e86522c29ac030e00977c1d4bce1/4-Figure1-1.png)
PDF] PHOTOCHEMICAL REDUCTION OF SILVER NITRATE TO NANO SILVER USING STANNOUS CHLORIDE, CTAB AND DAYLIGHT IRRADIATION | Semantic Scholar

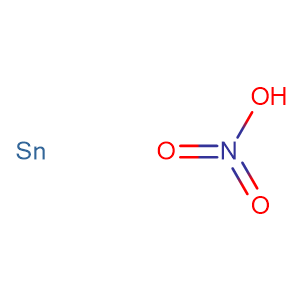
SOLVED: Namc Chem 3A All Naming - W oresEct Secuon Give the formulas for the following compounds ferric bicarbonate 2. sodium phosphite trisulfur pentachloride hydrogen gas 5 . stannous nitrate 6. hydrobromic
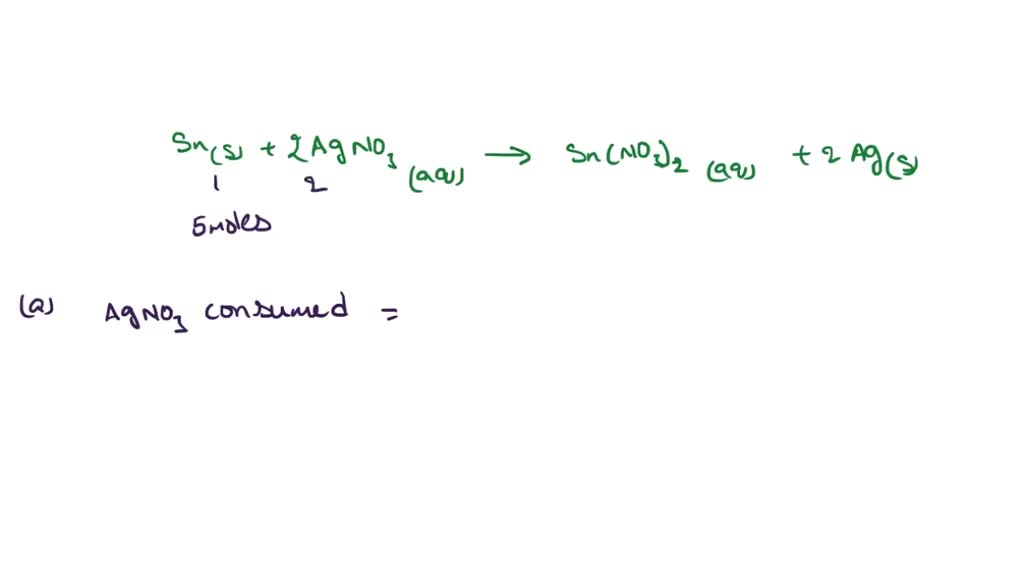
SOLVED: 3. Tin metal reacts with silver nitrate solution, according to: Sn(s) + 2AgNOs(aq) Sn(NO3), ( (aq) + 24g(s) For every .0 moles of Sn that reacts in this way: A How