Strategies for mitigating adverse events related to selective RET inhibitors in patients with RET-altered cancers: Cell Reports Medicine

Selpercatinib combination with the mitochondria-targeted antioxidant MitoQ effectively suppresses RET–mutant thyroid cancer | npj Precision Oncology

PDF) Selpercatinib combination with the mitochondria-targeted antioxidant MitoQ effectively suppresses RET–mutant thyroid cancer

Phase III study of selpercatinib versus chemotherapy ± pembrolizumab in untreated RET positive non-small-cell lung cancer | Future Oncology

Lilly Receives U.S. FDA Approval for Retevmo™ (selpercatinib), the First Therapy Specifically for Patients with Advanced RET-Driven Lung and Thyroid Cancers | Eli Lilly and Company
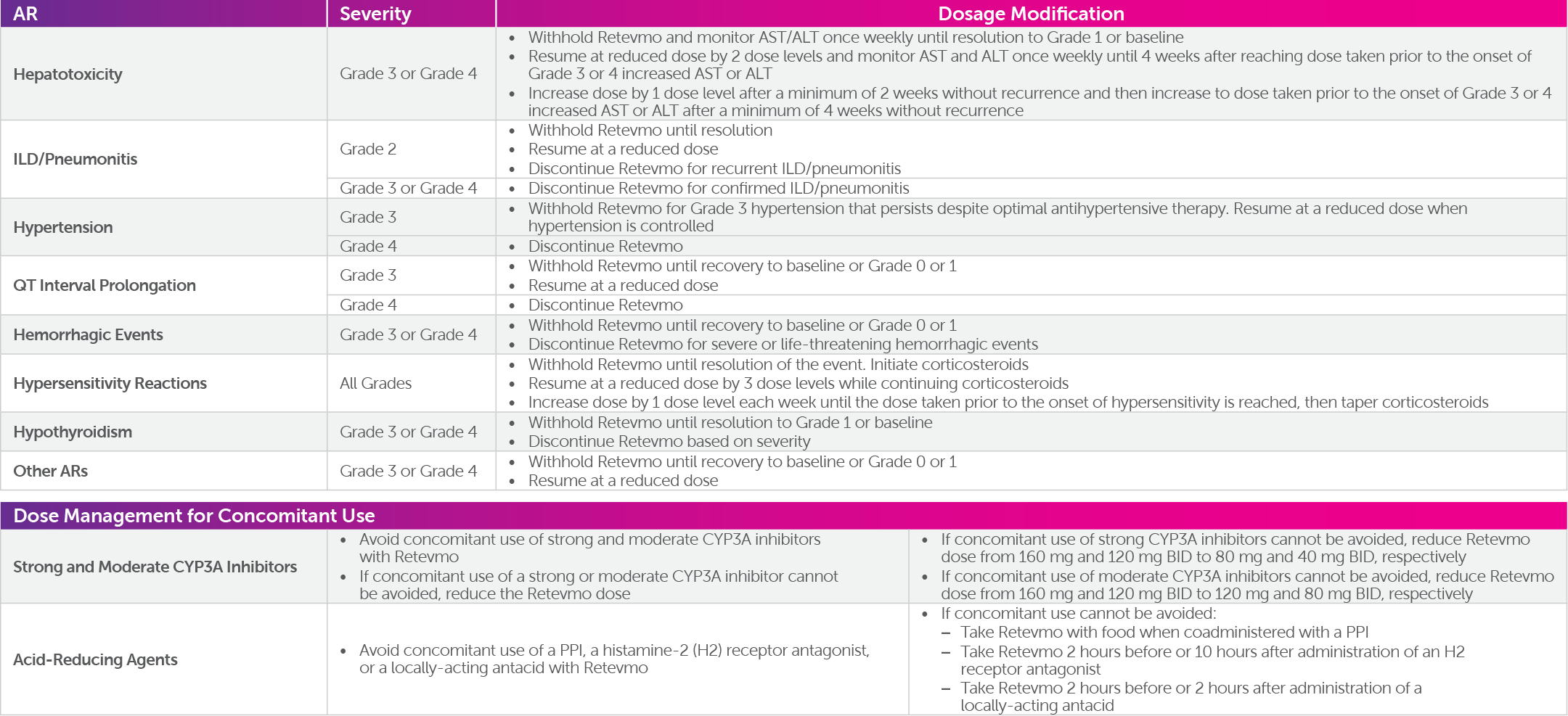
Written By: Joshua Nubla, PharmD NCODA Positive Quality Intervention: Selpercatinib (Retevmo®) Management Description: This PQI