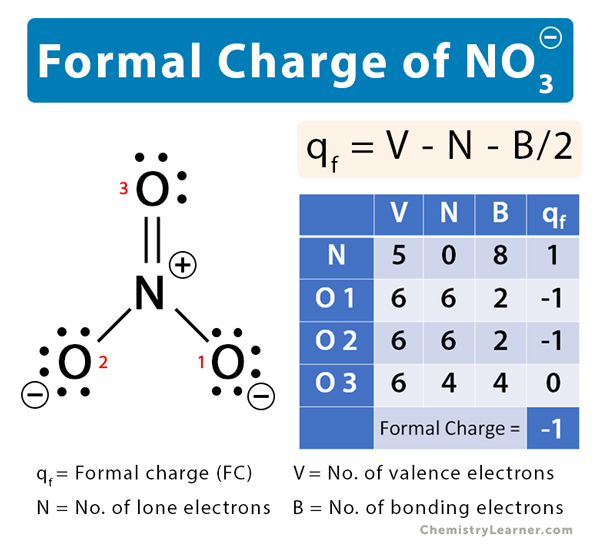
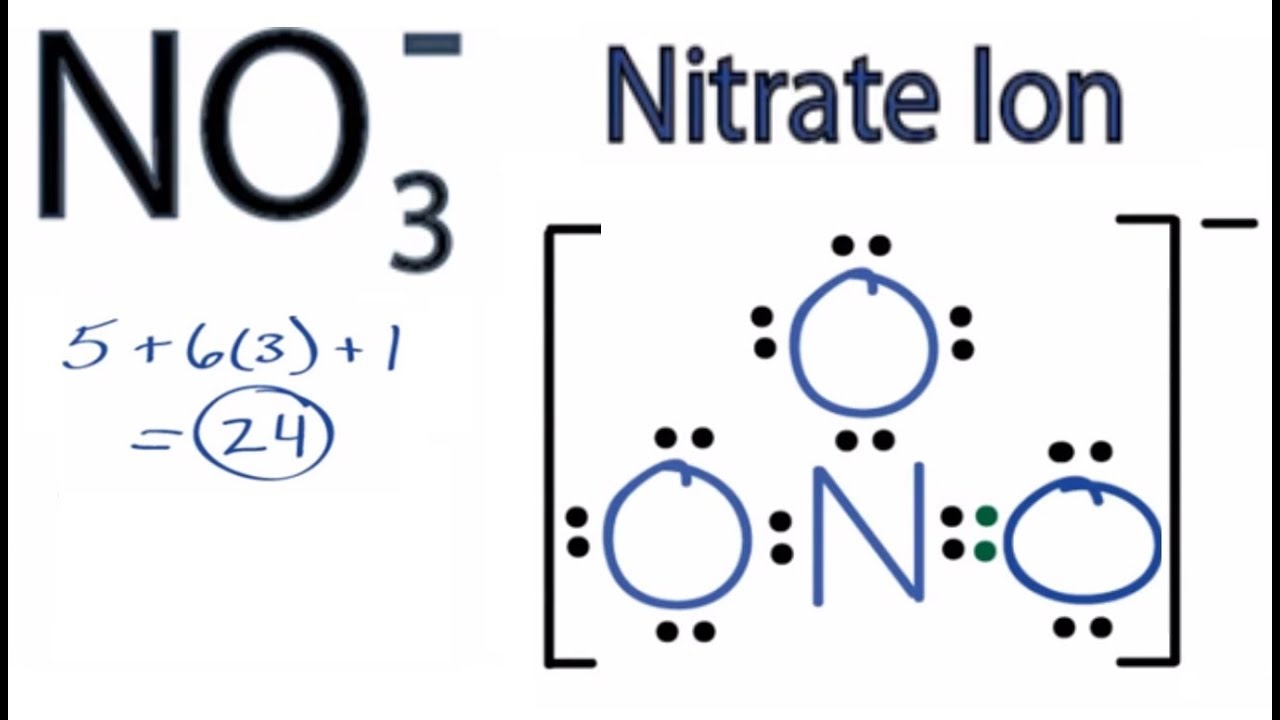
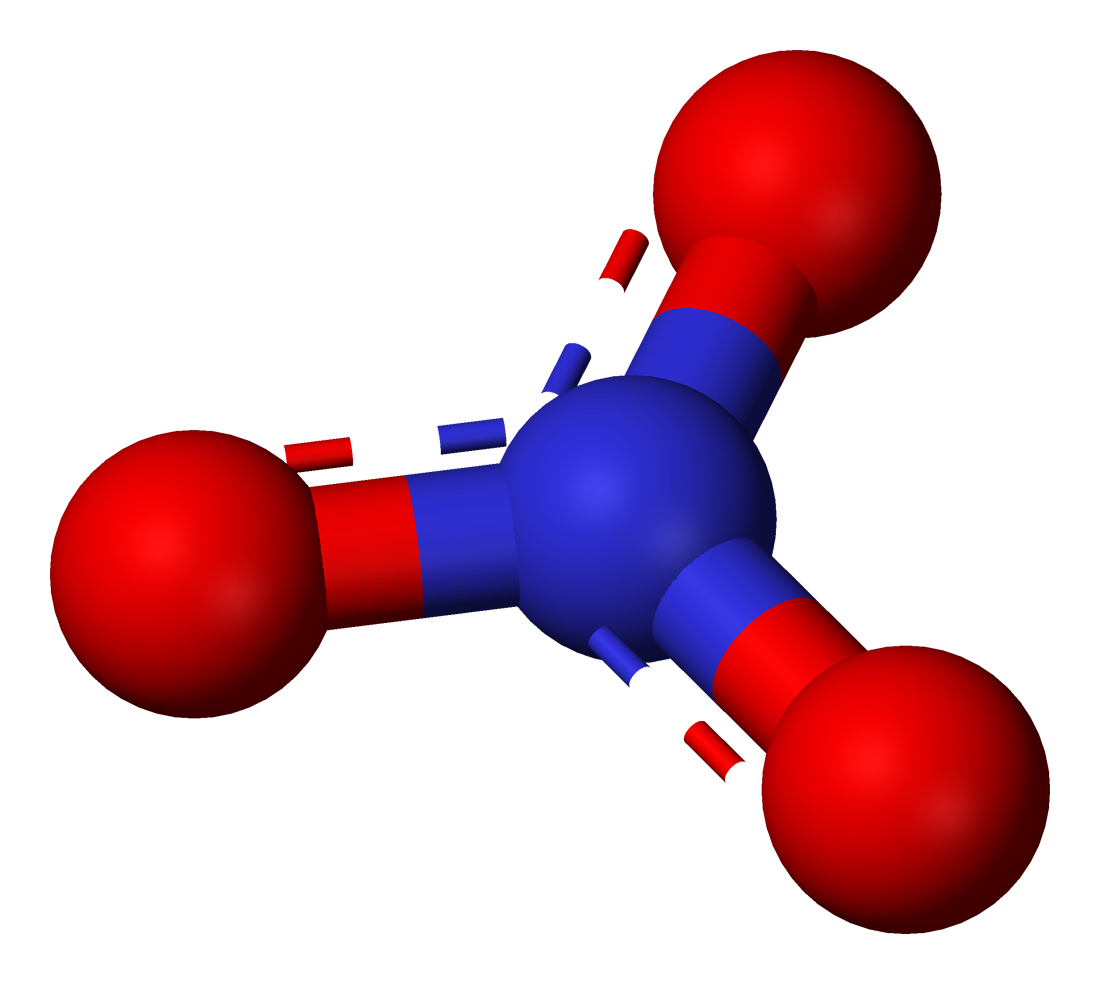
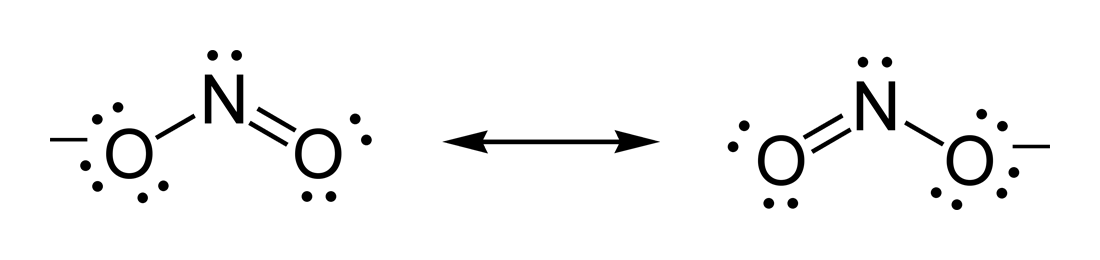
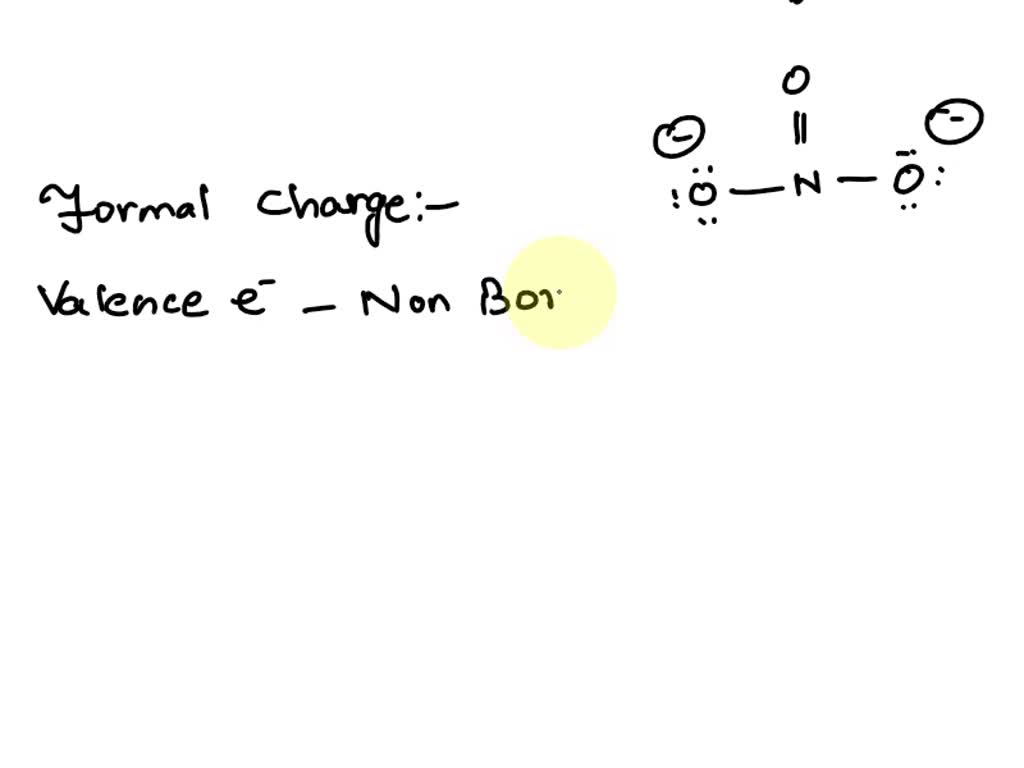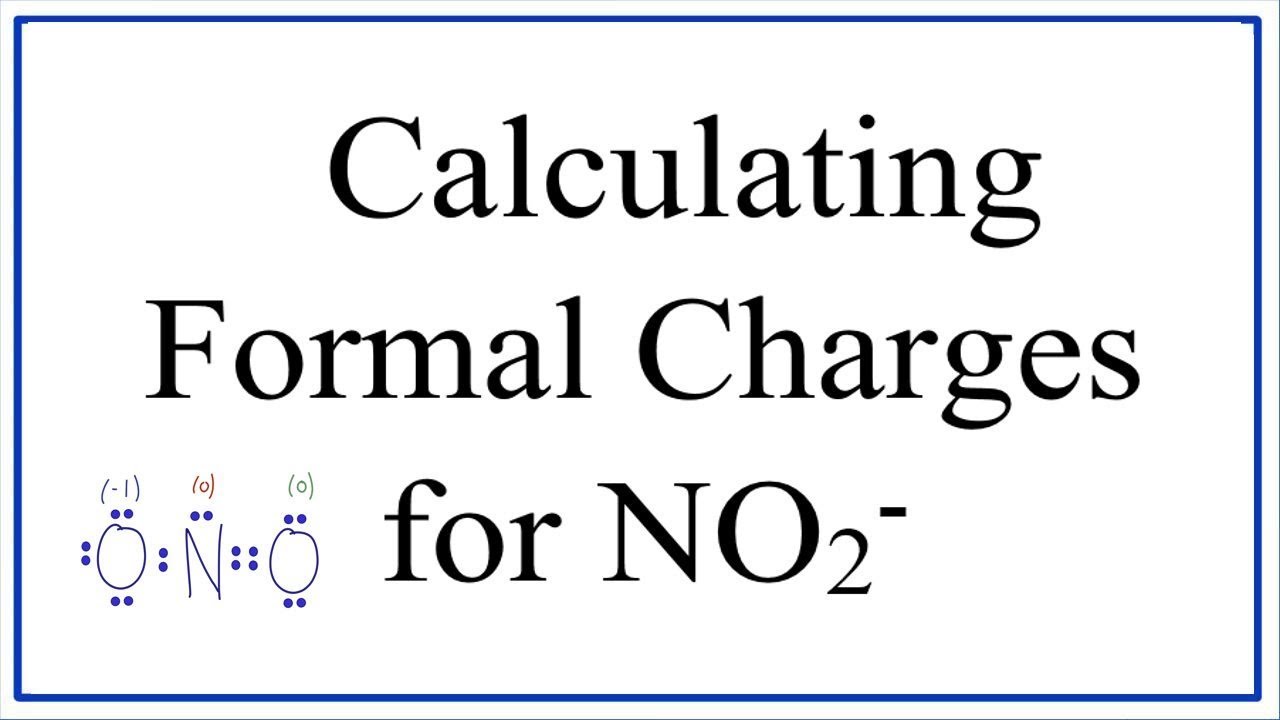39 Which of the following is true for nitrate anion (1) formal charge on N is 0 (2) bond order of NO bond is 4/3 (3) average formal charge on oxygen is
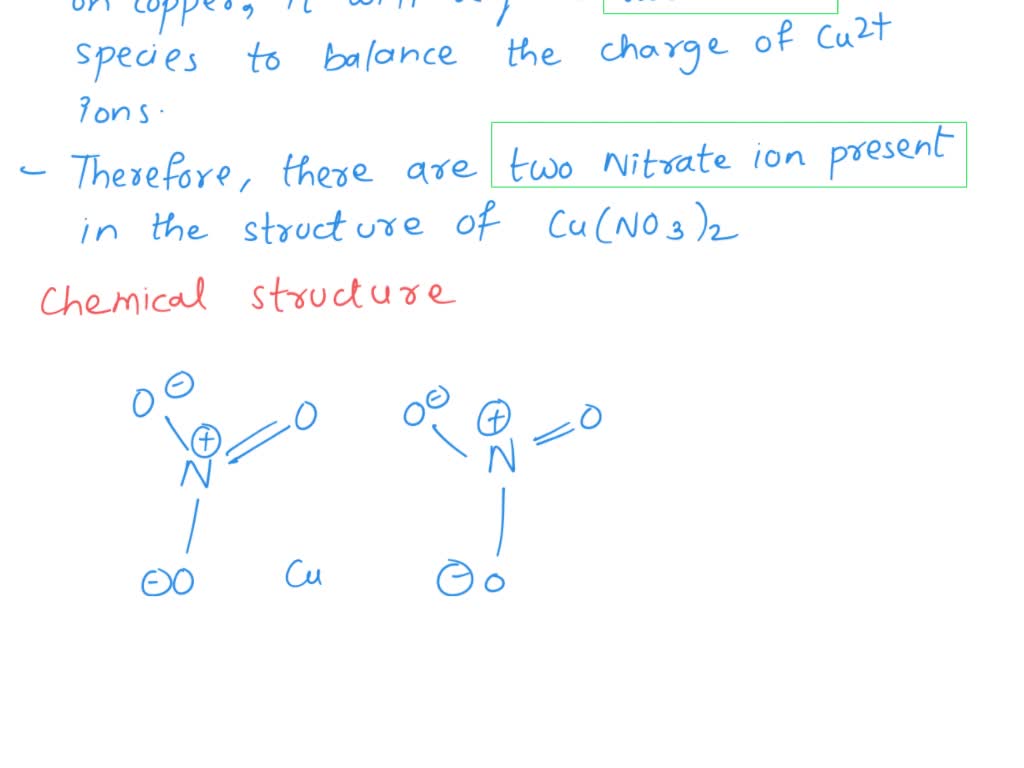
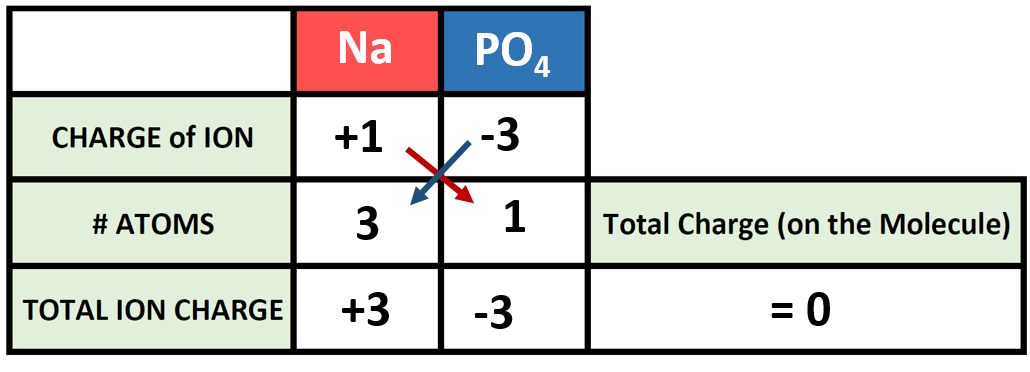
SOLVED: a) Why are there two nitrate ions in the structure of copper(II) nitrate, i.e. Cu(NO3)2? You should answer this question in terms of ionic charges. b) If you are looking at
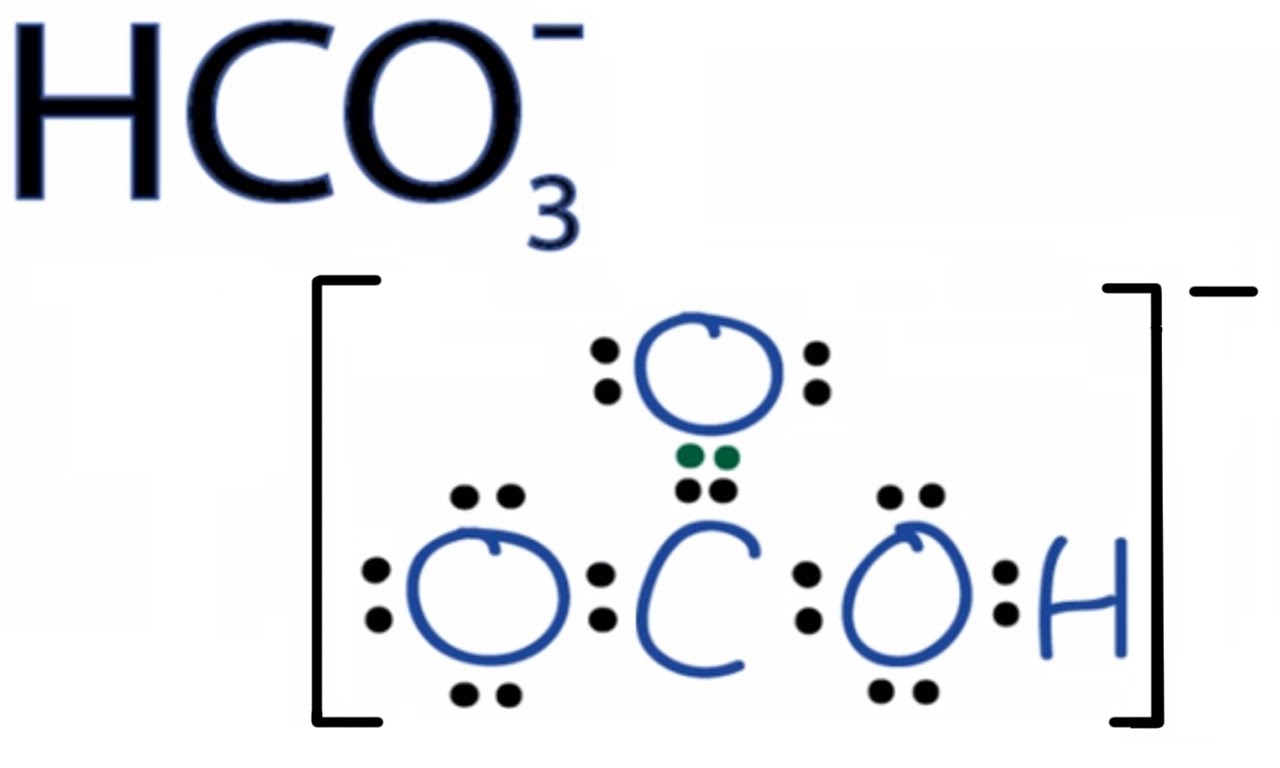
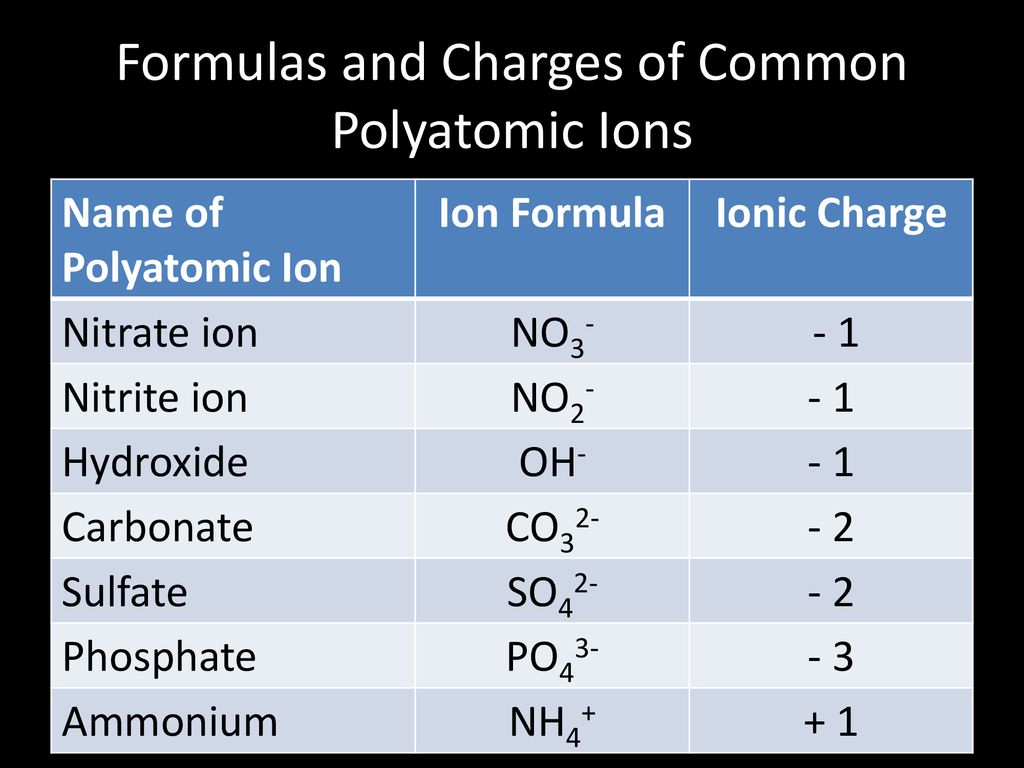
Which of the following polyatomic ions has a 3- ionic charge: hydrogen carbonate, hydroxide, sulfate, nitrate, or phosphate? | Socratic