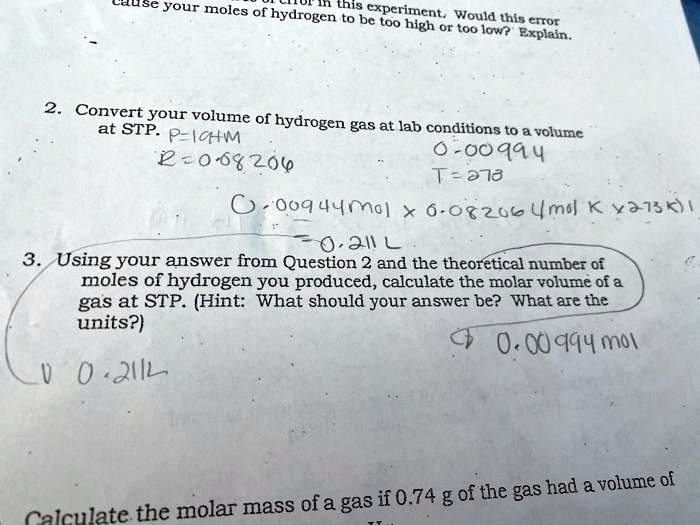Sample Lab Report Molar volume of Hydrogen - Sample Lab Report: Molar Volume Of Hydrogen Background: - Studocu

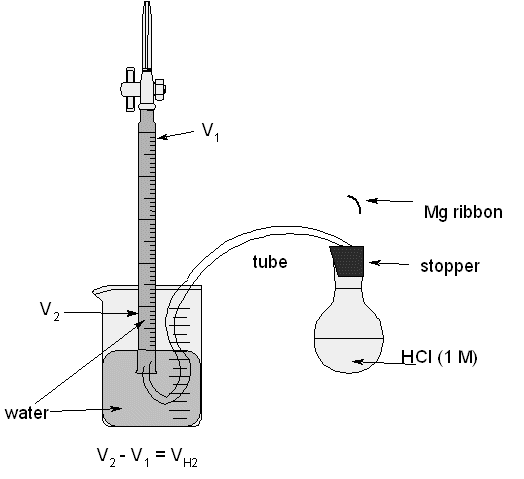
Sample Lab Report Molar volume of Hydrogen - Sample Lab Report: Molar Volume Of Hydrogen Background: Gasses are a special phase of matter that can be | Course Hero
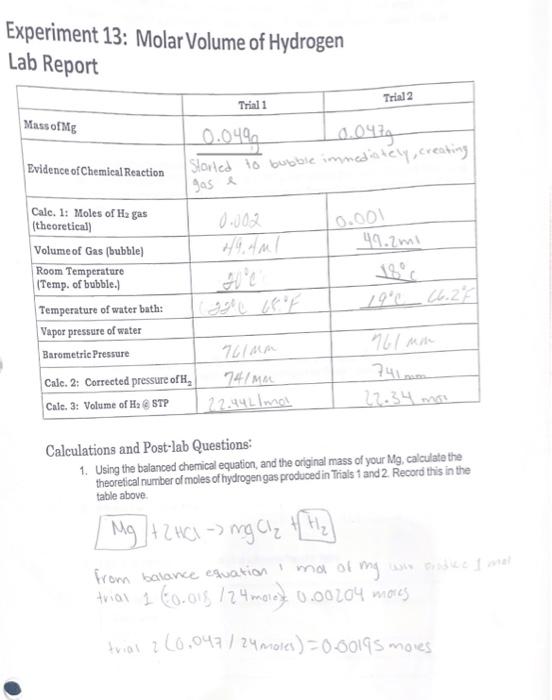
Solved: Molar Volume of Hydrogen Reaction of 0.028 g of magnesium with excess hydrochloric acid g [Chemistry]

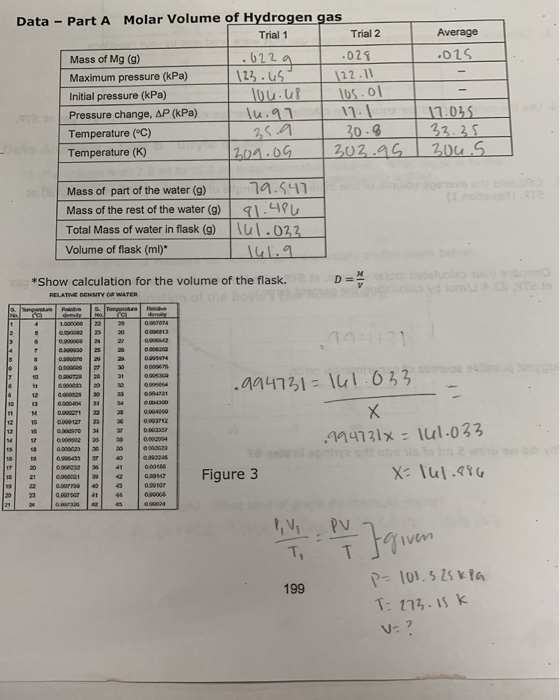
SOLVED: Molar Volume of Hydrogen Data Table Trial 1 Trial 2 X 009439 X a.H8mL 8 .28-L 22.8 C Length of Mg Ribbon Mass of Mg 00942 g Evidence of Chemical Reaction

SOLVED: A student performed an experiment to determine the molar volume of hydrogen using the reaction between magnesium and excess hydrochloric acid. The following data was collected: - Room temperature: 21°C -