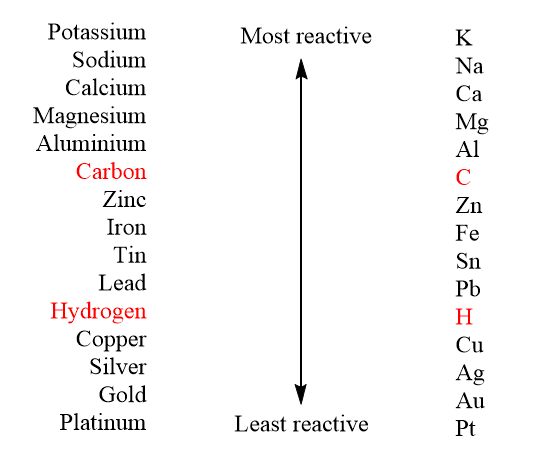
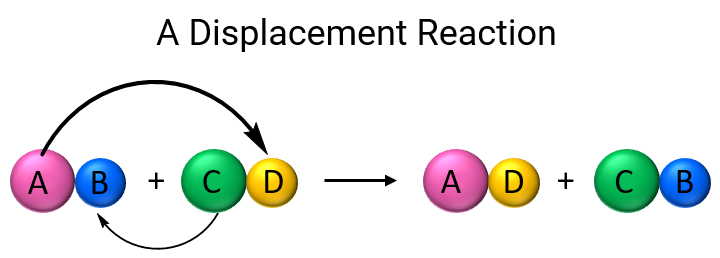
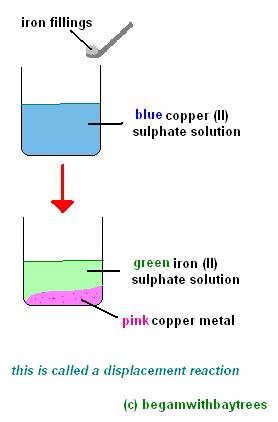
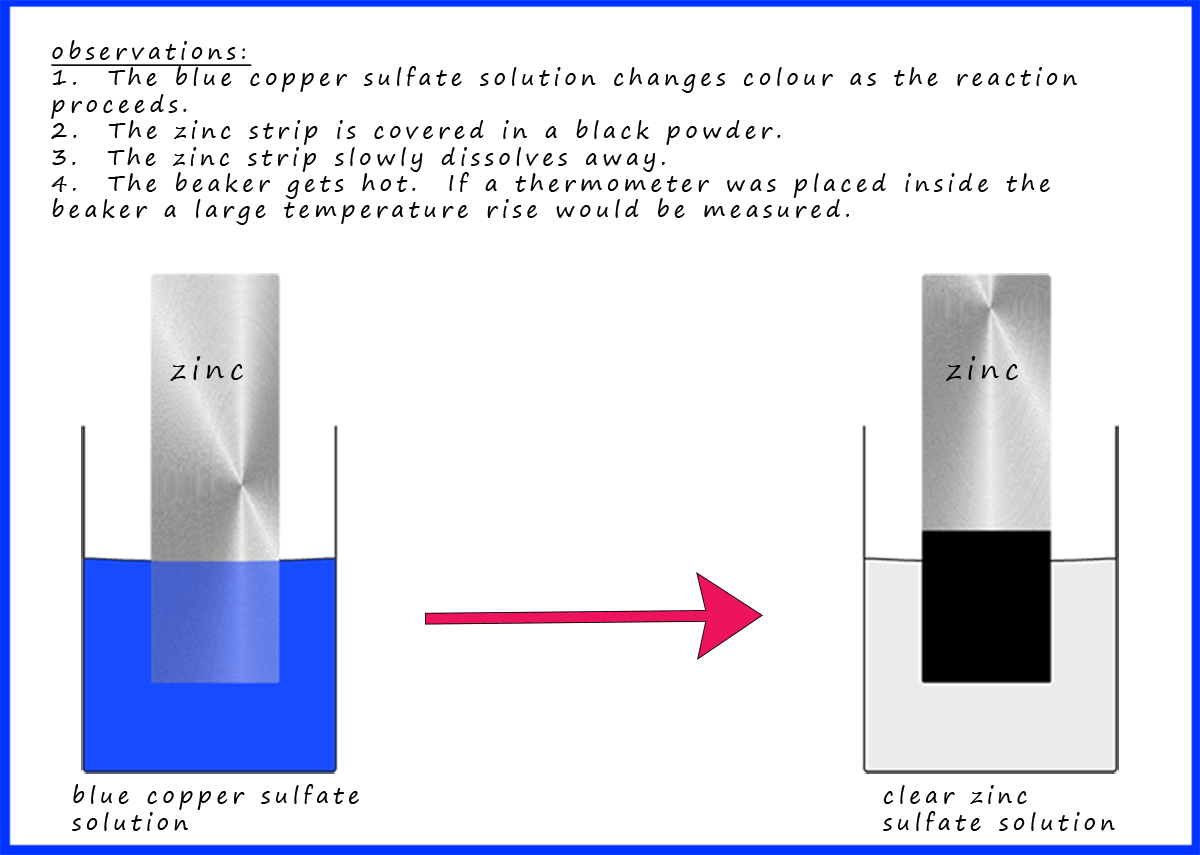
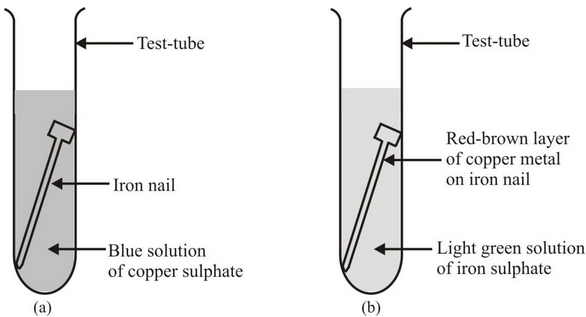
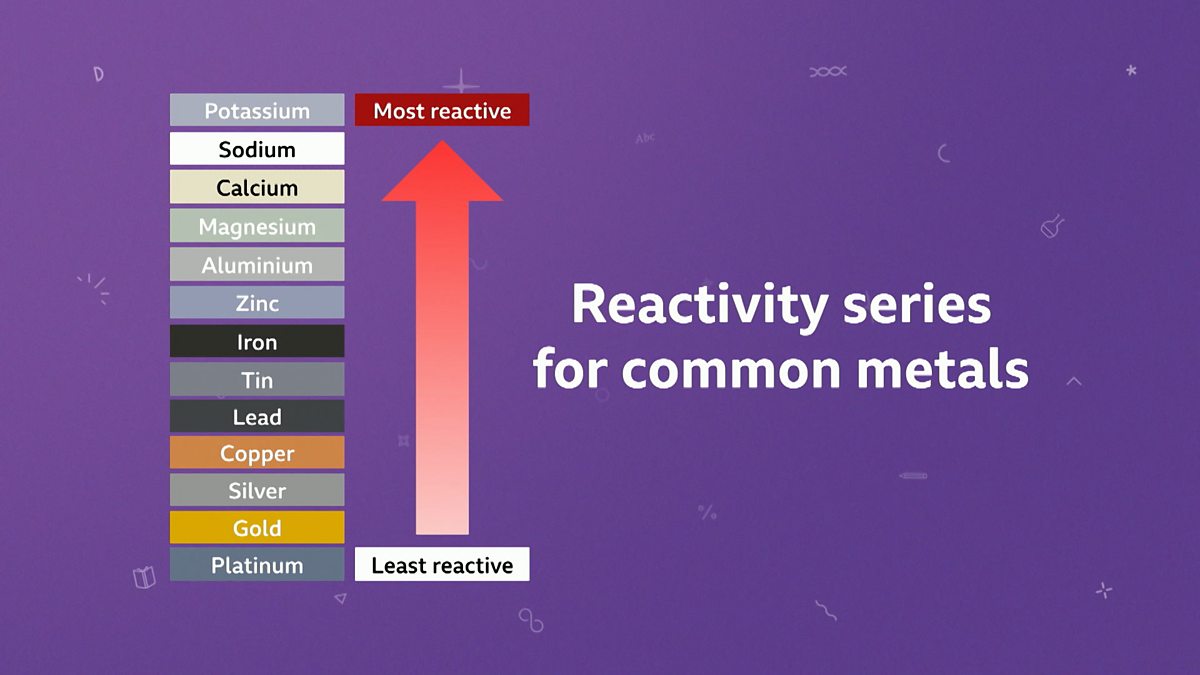
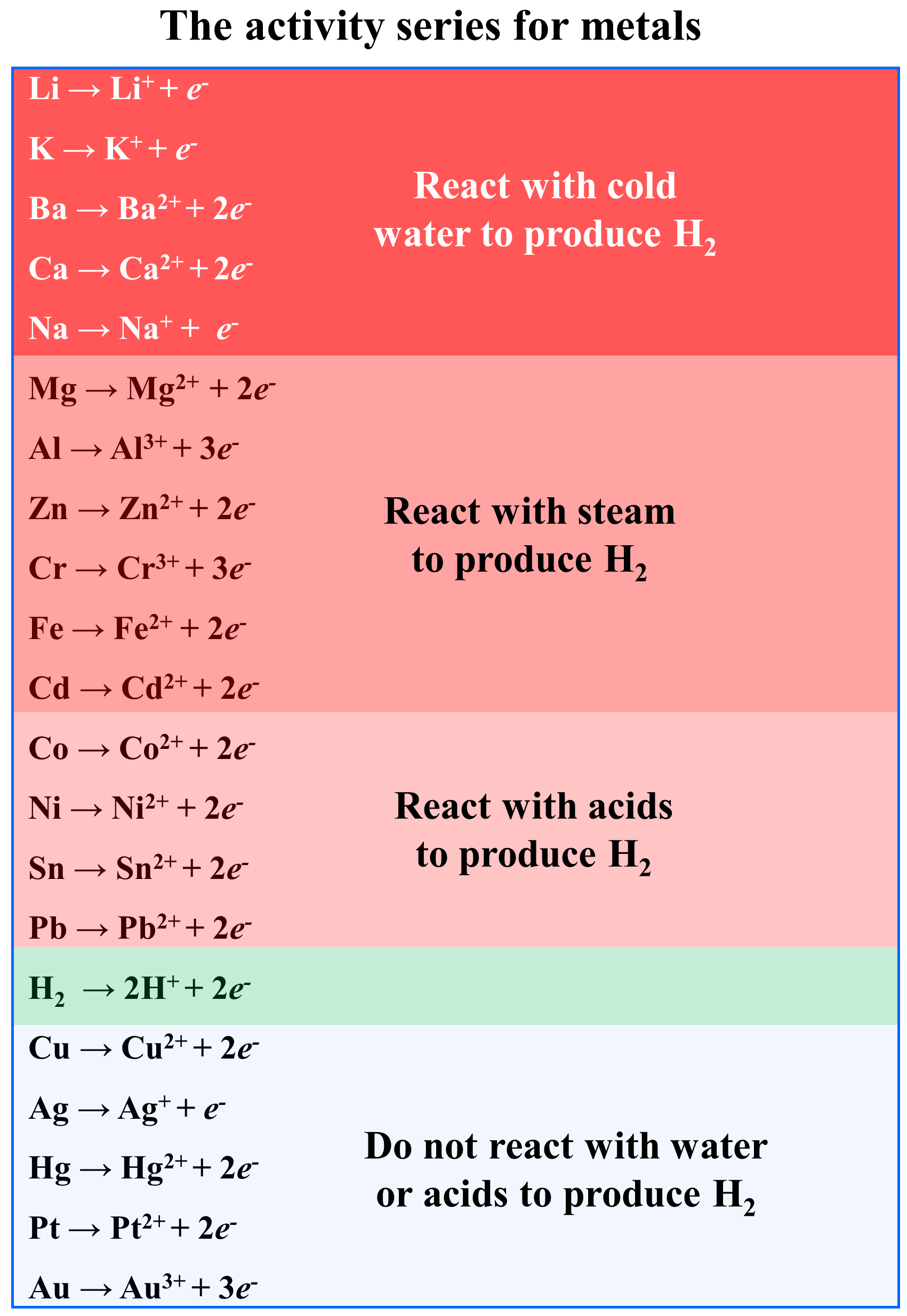savvy-chemist: GCSE OCR Gateway C4:1d-f Displacement reactions of metals: The Reactivity Series of Metals (2)

please explain the displacement reaction of metals and non-metals - Chemistry - Redox Reactions - 4575986 | Meritnation.com

Metal reactivity series experiments results observations conclusions deducing reactivity order gcse igcse KS4 science chemistry revision notes revising O level notes
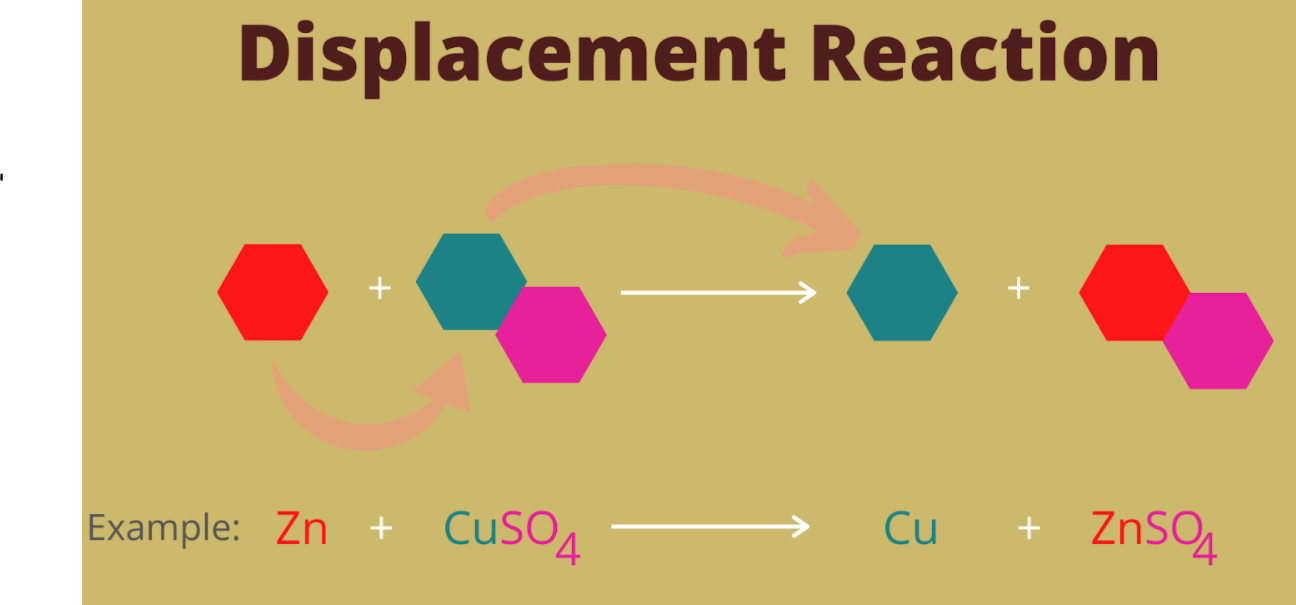
Which displacement reaction will not occur among the following reactions?A) ${ Zn+ } { CuSO }_{ 4 }$B) ${ Fe+ } { CuSO }_{ 4 }$C) ${ Cu+ } { FeSO }_{ 4 }$D) ${ Mg+ } { CuSO }_{ 4 }$