Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 7-10 November 2022

Medicinal products for human use - Formalities & procedures - Single Window for Logistics - Luxembourg
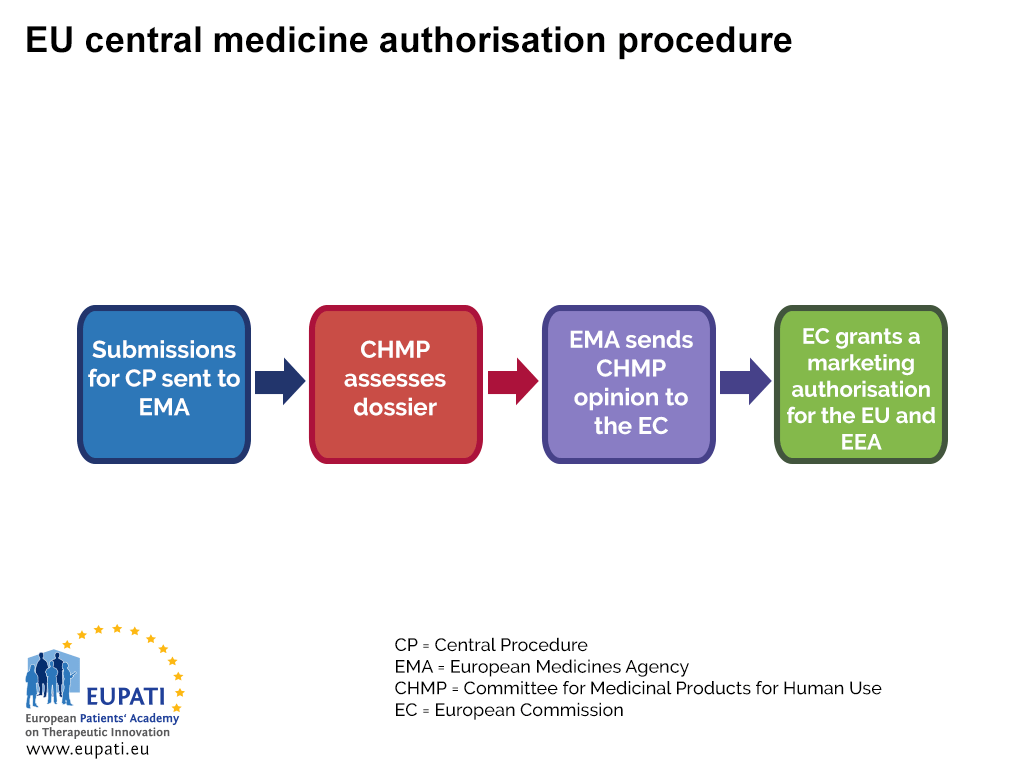
Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 18-21 March 2024 | European Medicines Agency

Medicinal products in the European Union: The legal framework for medicines for human use | Epthinktank | European Parliament

Italy: AIFA Guidelines on marketing authorization application for Medicinal Products for Human Use - Global Compliance News

GMP_MANUFACTURE OF BIOLOGICAL MEDICINAL PRODUCTS FOR HUMAN USE - EU GMP- ANNEX 2- VOL IV by Cryolab - Issuu