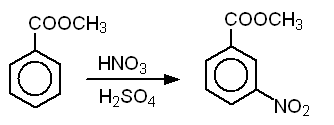
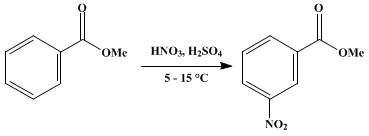
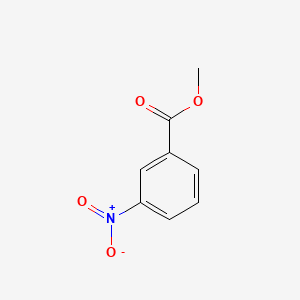
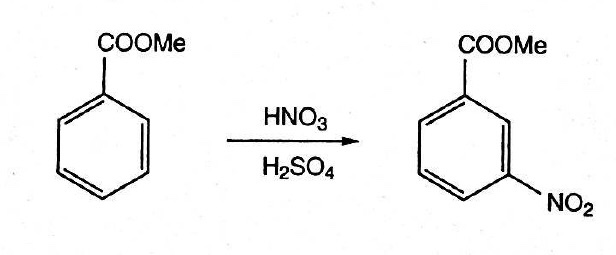
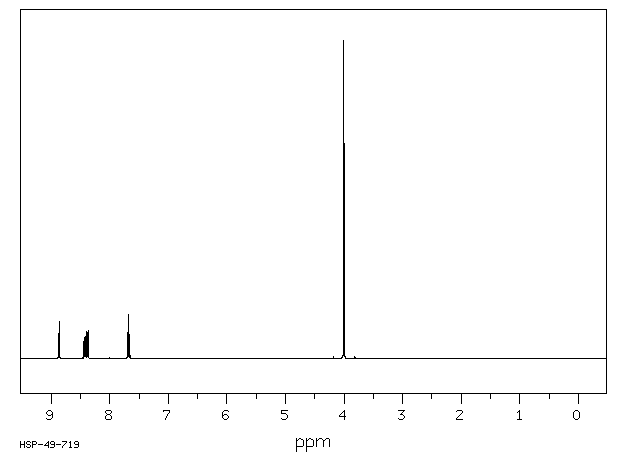
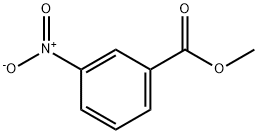
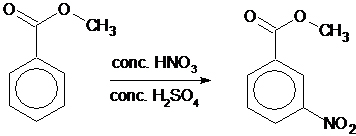Why is m-nitro formed in the reaction of nitration of methyl benzoate instead of the ortho or para isomers? Draw the structure of the products formed on nitration of each of the
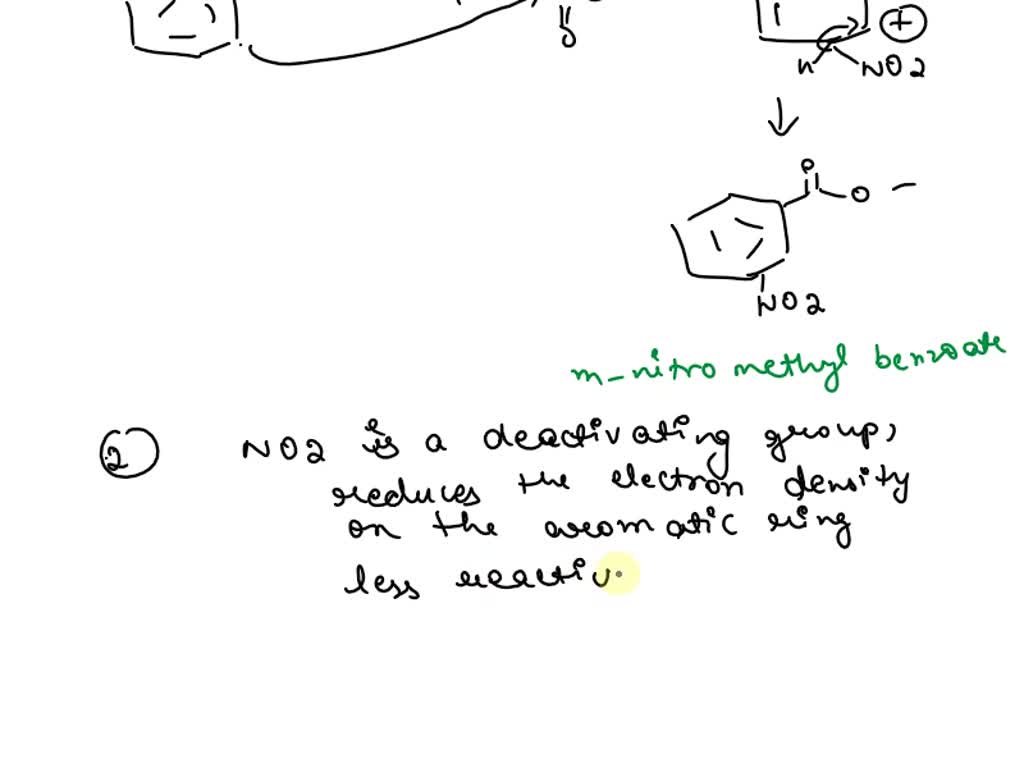
SOLVED: Although methyl m-nitrobenzoate is the principal product formed in the reaction, impurities will include small amounts of the ortho and para isomers of methyl nitrobenzoate and the dinitration product. These side

PDF) NITRATION OF METHYL BENZOATE (ELECTROPHILIC AROMATIC SUBSTITUITION | idayu razali - Academia.edu
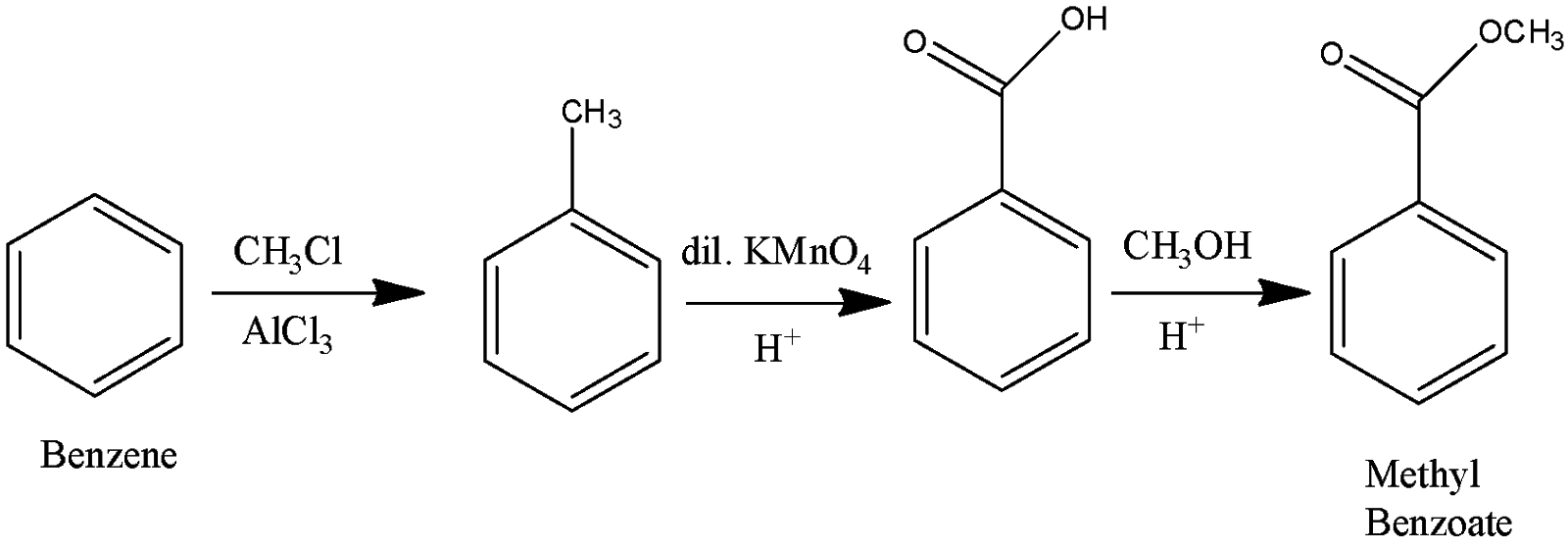
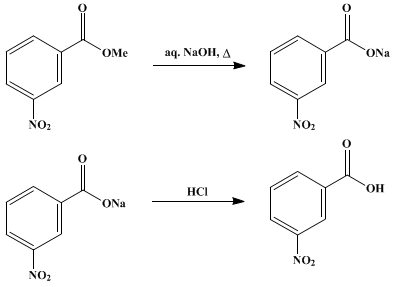
How will you prepare the following compounds from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom:i. Methyl benzoate ii. m-Nitrobenzoic acidiii. p-Nitrobenzoic

Write the mechanism of the reaction of methyl benzoate to form methyl 2-nitrobenzoate. - brainly.com