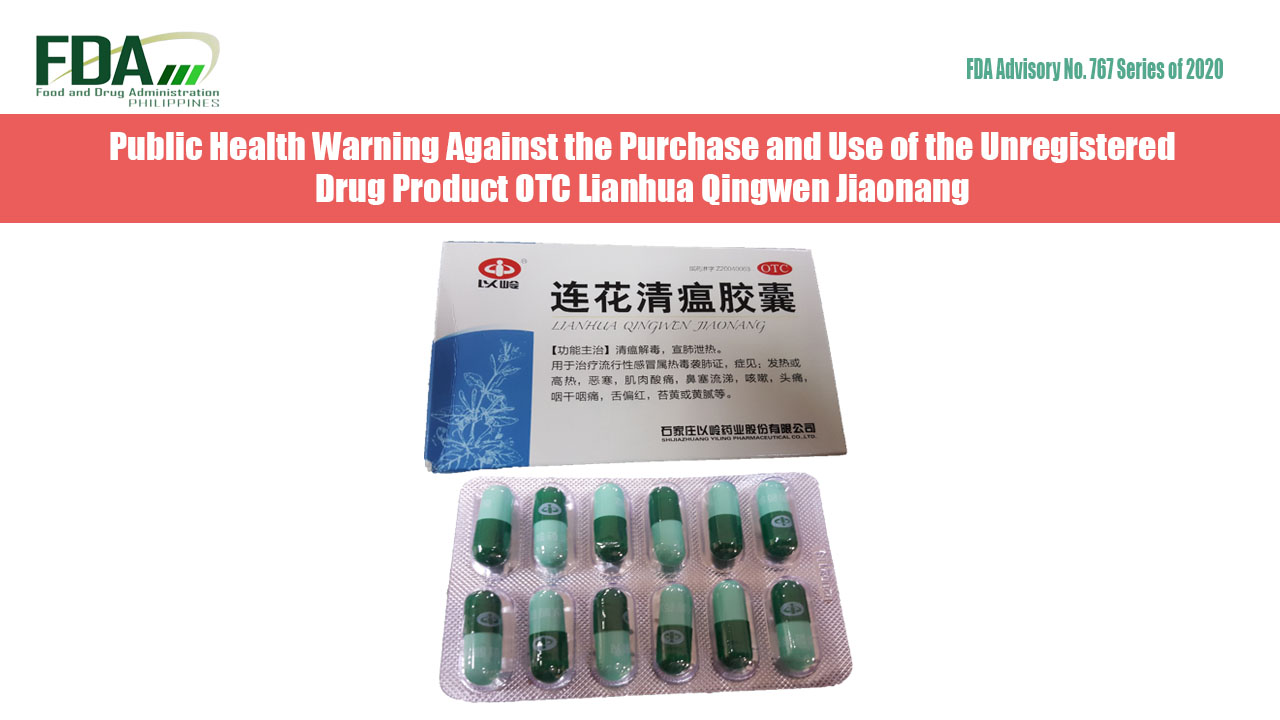
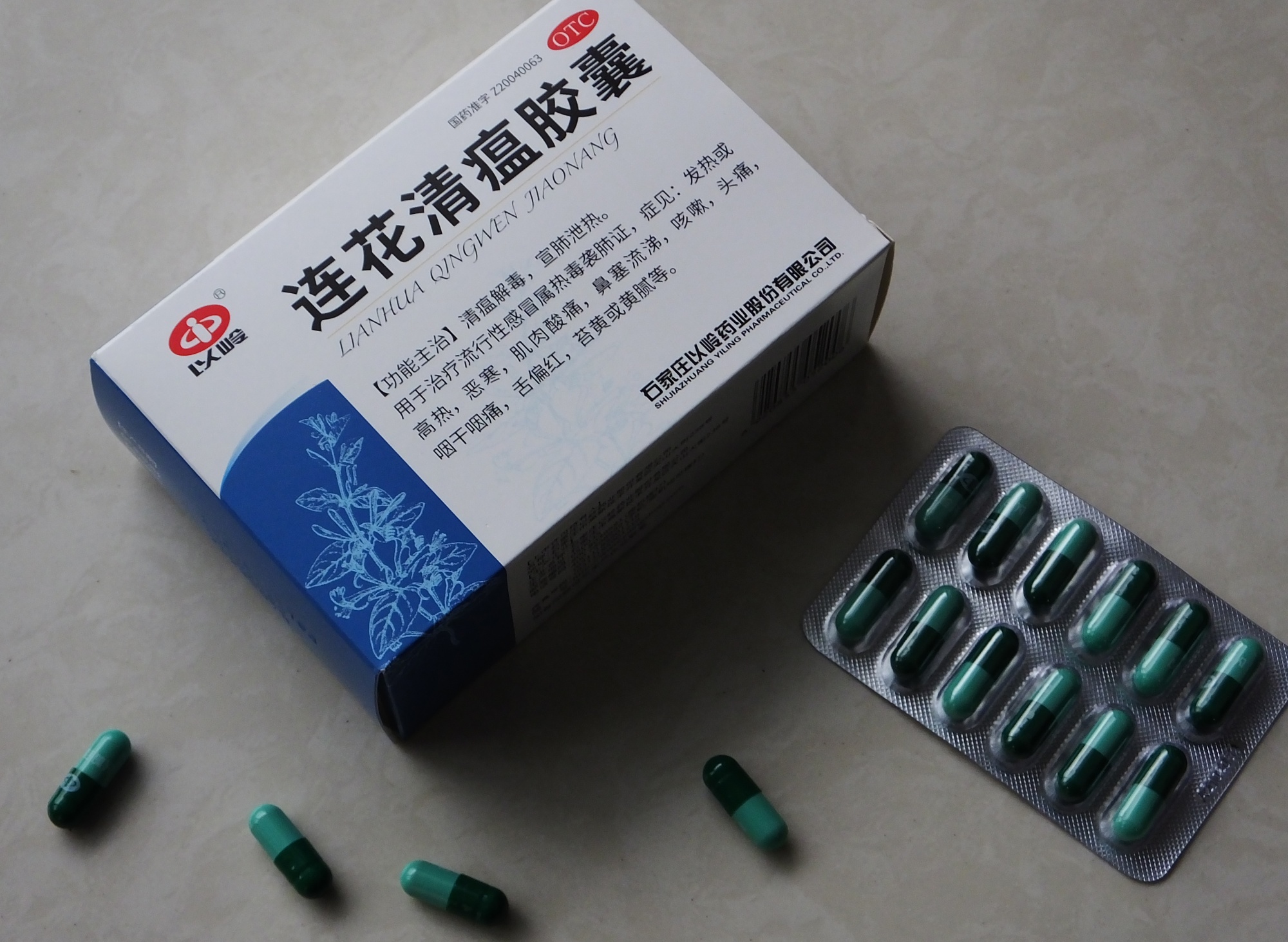
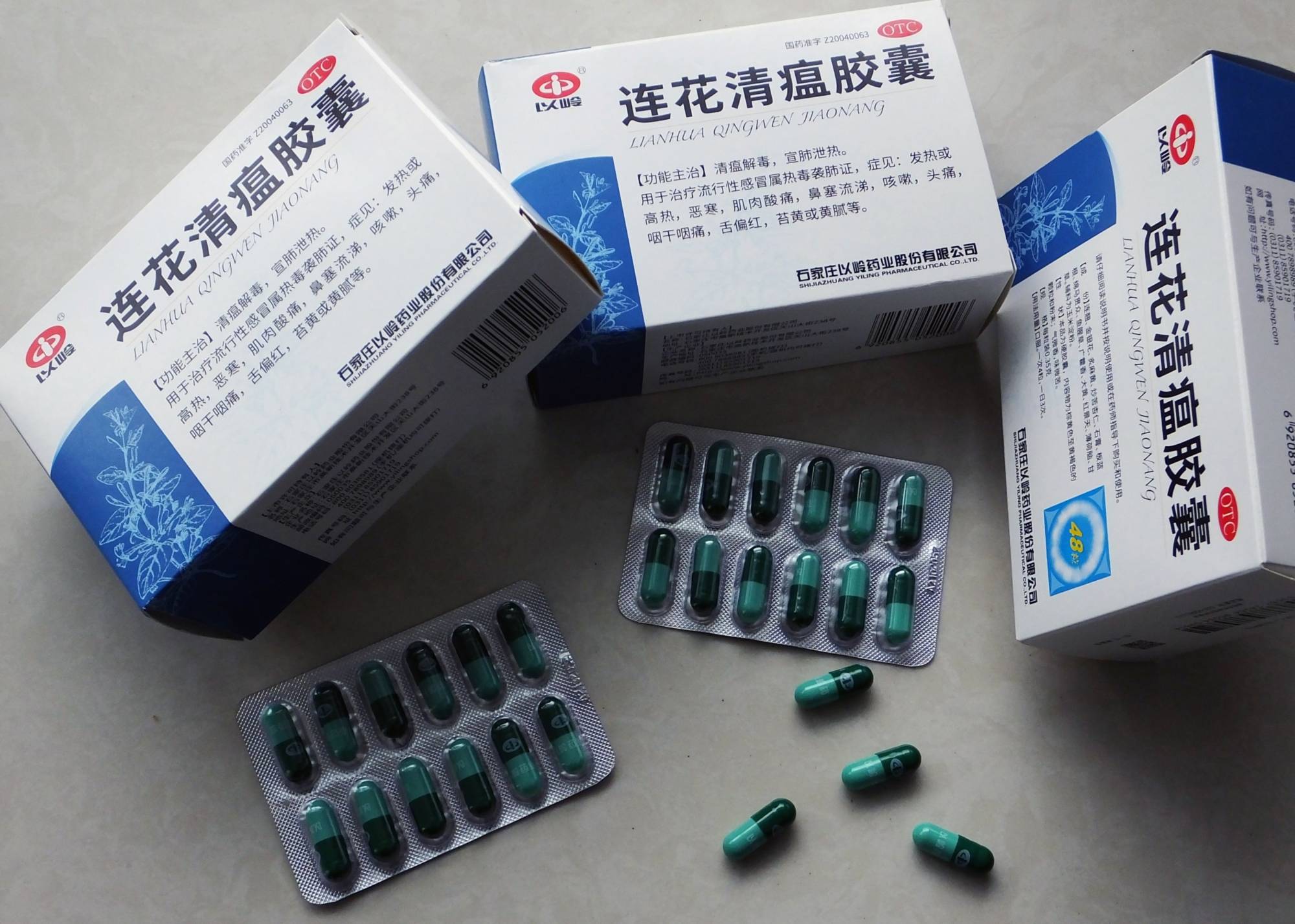
FDA Advisory No.2021-0723 || Public Health Warning Against the Continuous Selling of the Unregistered Drug Product Lianhua Qingwen Jiaonang with Chinese Characters - Food and Drug Administration

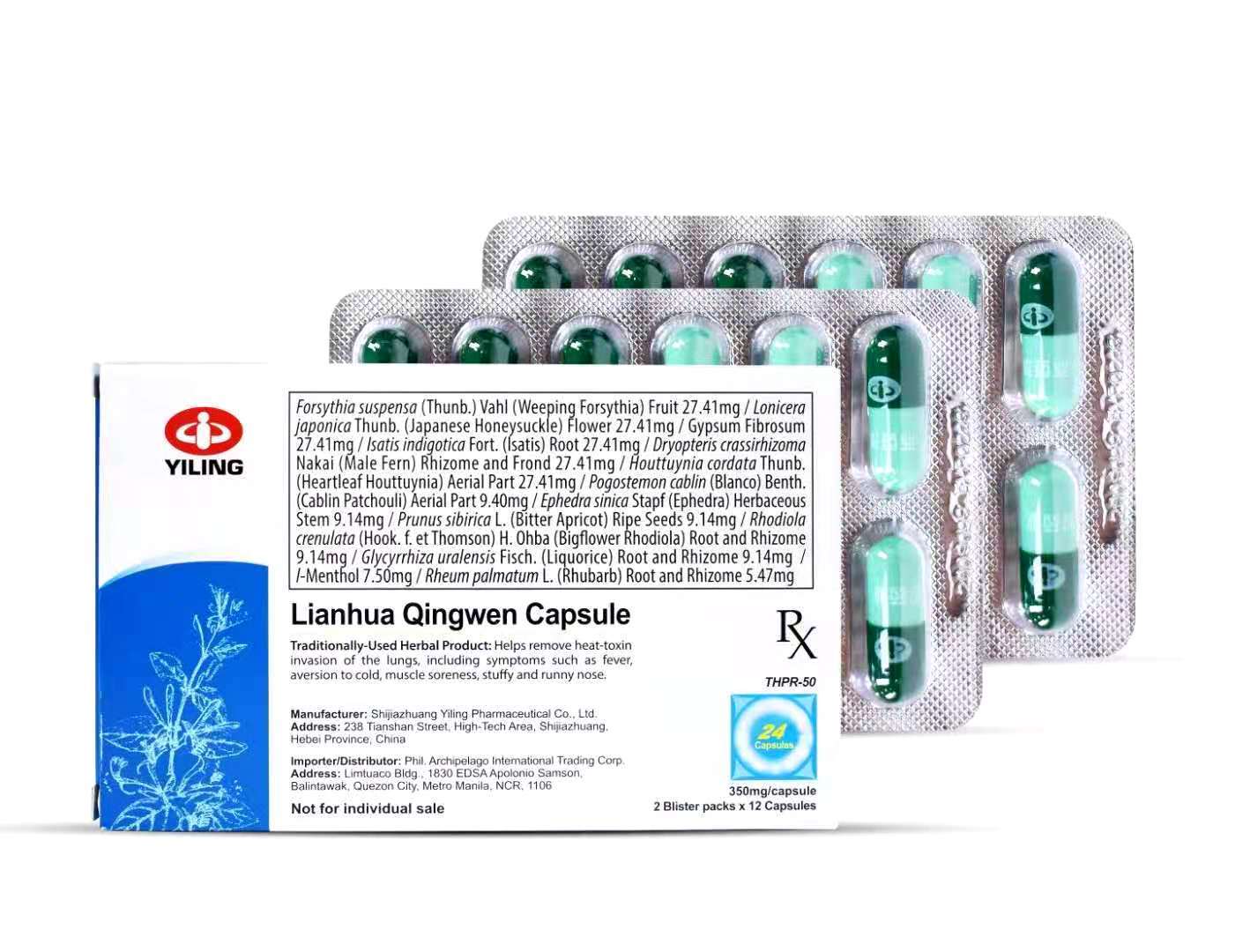
FDA Advisory No.2022-1612-A || Babala sa Publiko Tungkol sa Pagbili at Paggamit ng Hindi Rehistradong Gamot na “Lianhua Qingwen Capsules 24's” 1. - Food and Drug Administration
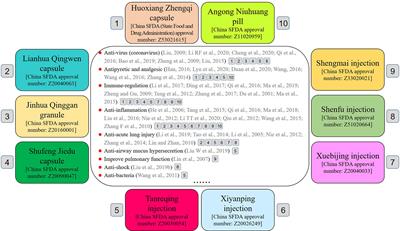
Frontiers | Chinese Patent Medicines in the Treatment of Coronavirus Disease 2019 (COVID-19) in China

Lianhua Qingwen prescription for Coronavirus disease 2019 (COVID-19) treatment: Advances and prospects - ScienceDirect