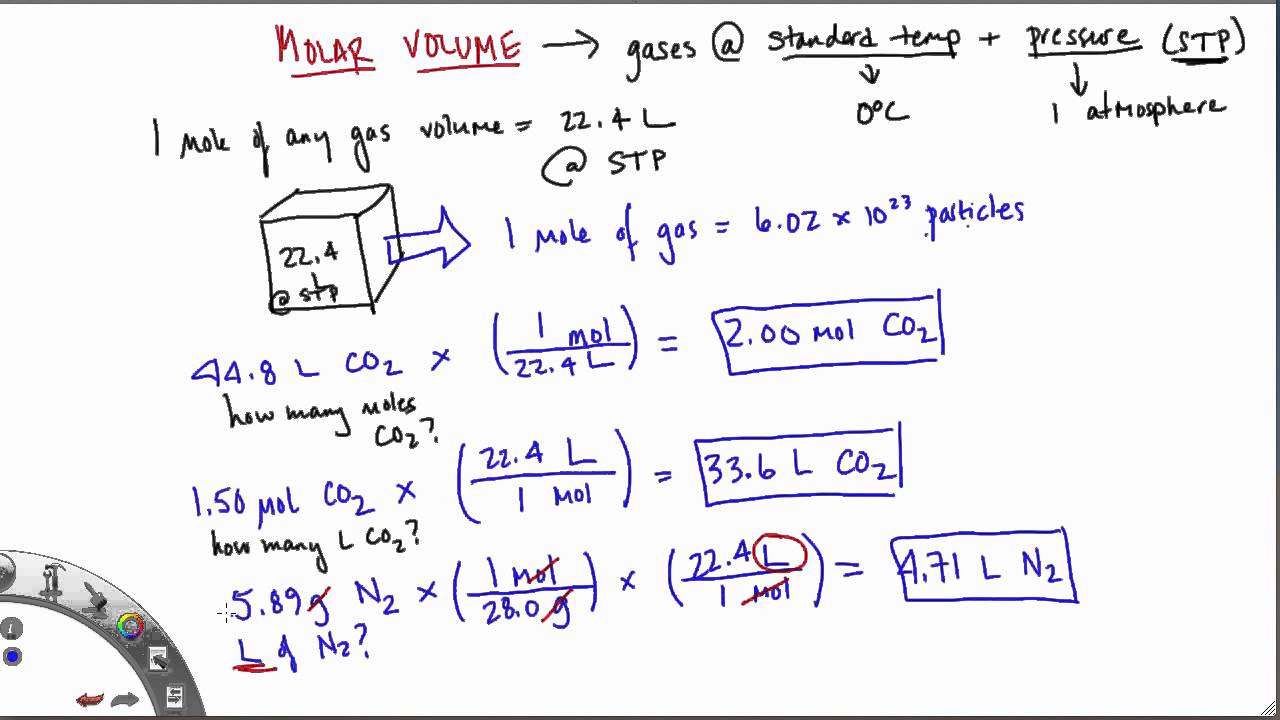
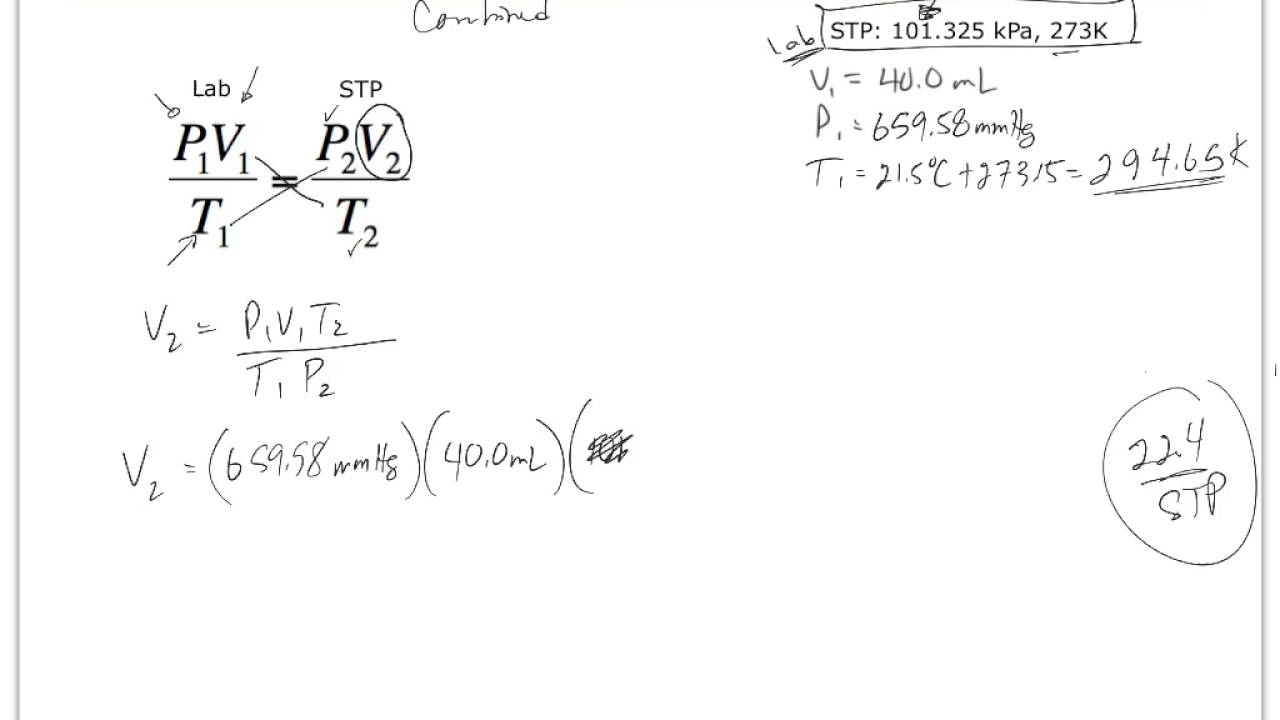Mole and gas volume The molar volume of a gas is its volume per mole, litre mol-1. It is the same for all gases at the same temperature and pressure. The. -

SOLVED: Use molar volume to calculate each of the following at STP: a. the number of moles of O2 in 44.8 L of O2 gas b. the volume, in liters, occupied by

Mole and gas volume The molar volume of a gas is its volume per mole, litre mol-1. It is the same for all gases at the same temperature and pressure. The. -

SOLVED: Use molar volume to calculate each of the following at STP: a. the volume, in liters, of 6.40 g of O2 gas b. the number of grams of H2 in 1620
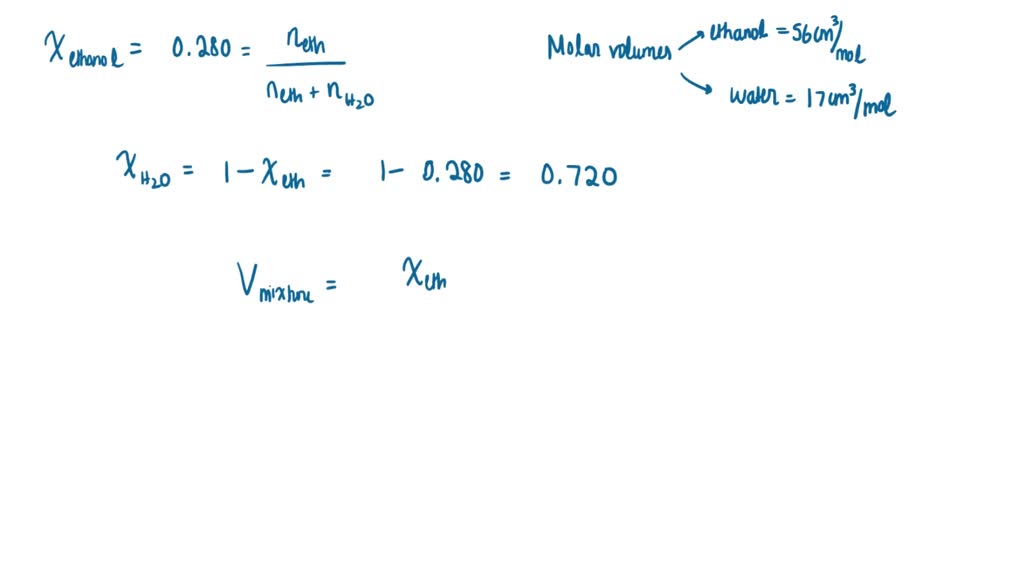
SOLVED: Calculate the volume of one mole (molar volume) of a mixture of water and ethanol in which mole fraction of ethanol is 0.280. Partial molar volumes of ethanol and water at