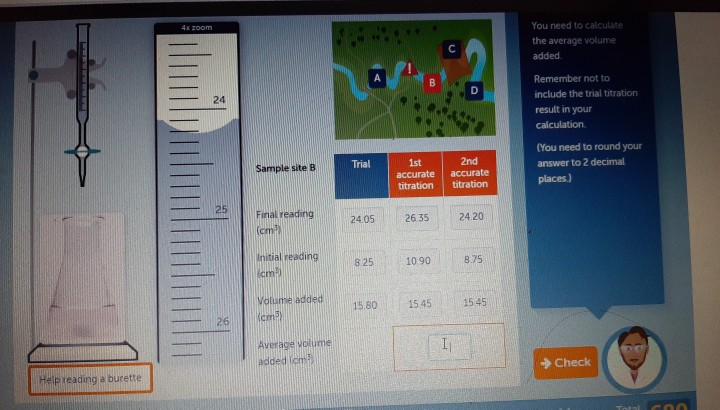
SOLVED: Lab Data Liquid Level Measurement Water Volume 030 000 mL = Liter Volume of Drop Size Number of Drops Average Drop Size (mL) 0.05 average drop size (mL/number of drops) Volume

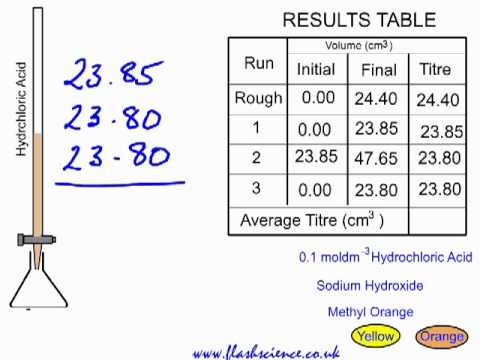
SOLVED: Data and Observations: Fill in Table 1 below using the data provided at the end of the video. To calculate the volume of AgNO3 used, subtract the initial reading from the
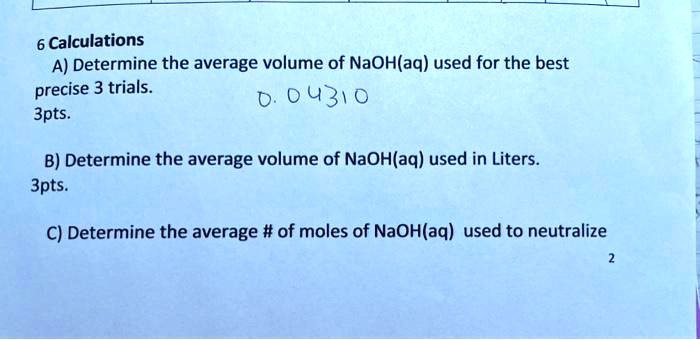
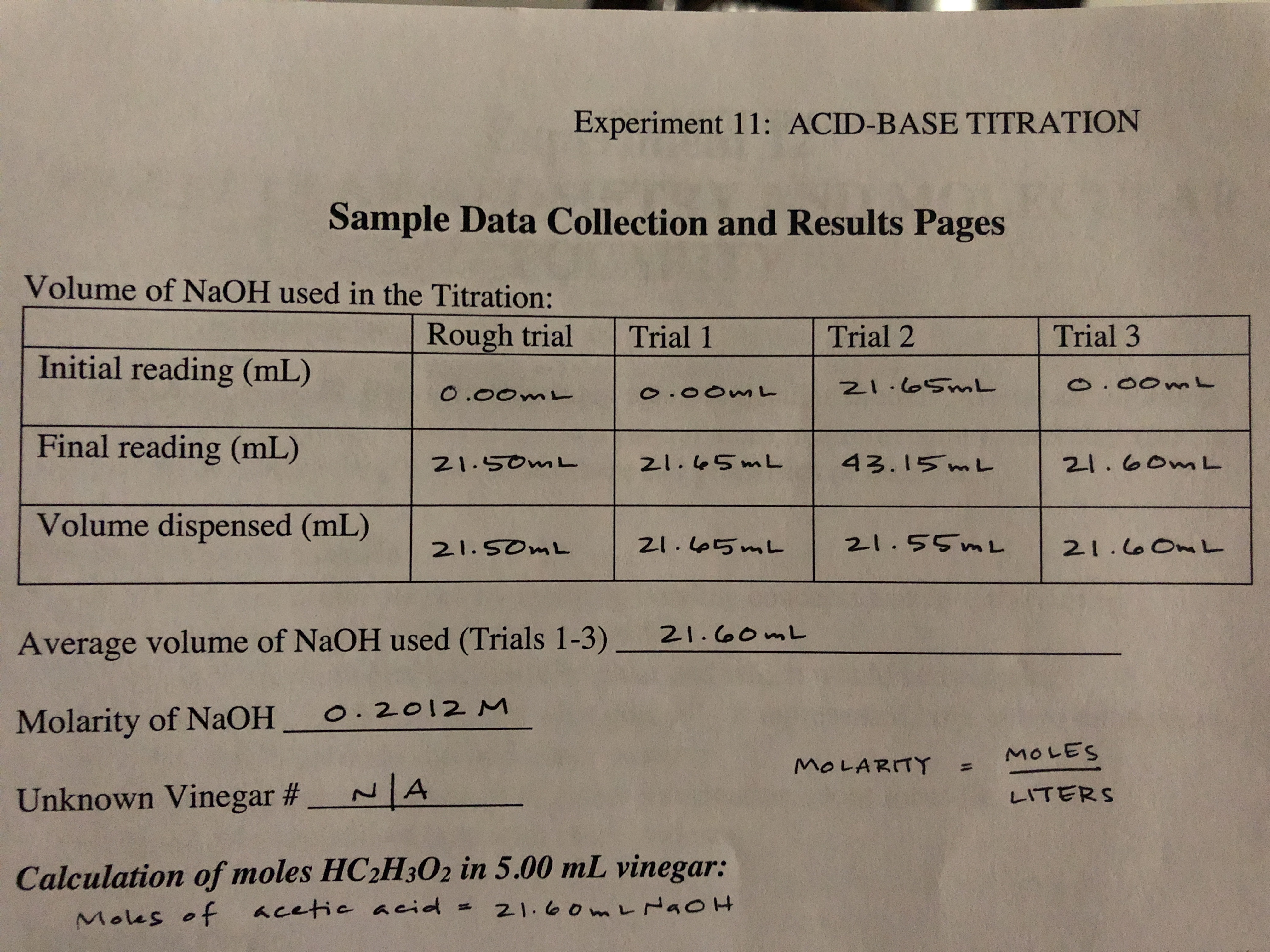
SOLVED: Calculations A) Determine the average volume of NaOH(aq) used for the three most precise trials. 4.31 L B) Determine the average volume of NaOH(aq) used in liters. 4.31 L C) Determine

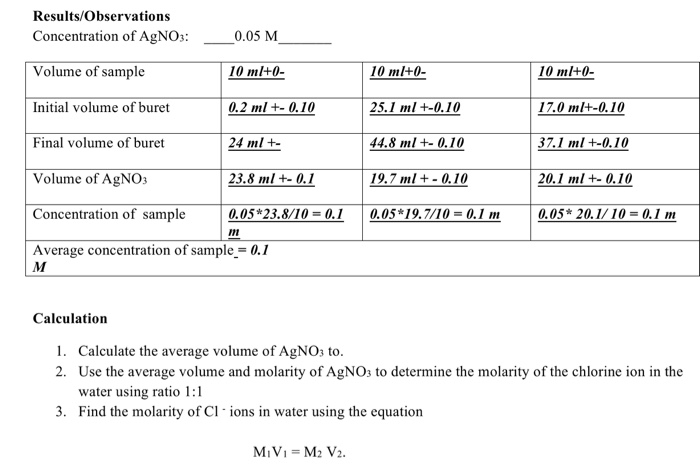






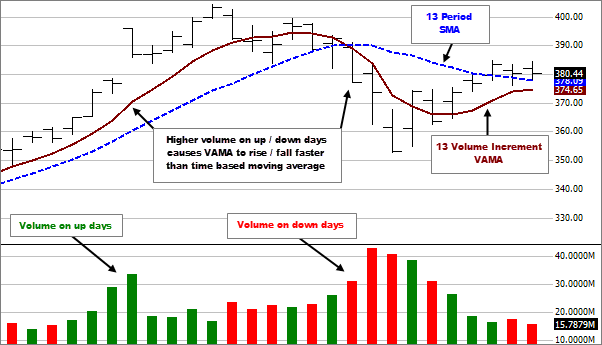

:max_bytes(150000):strip_icc()/Volume-Weighted-Average-Price-VWAP-FINAL-661fcb9d30a04362a4345e33e6b73183.png)