Hydrogen Peroxide I.P 6% 20 Volumes at Rs 35/litre | GACL Hydrogen Peroxide in Pune | ID: 1837285488
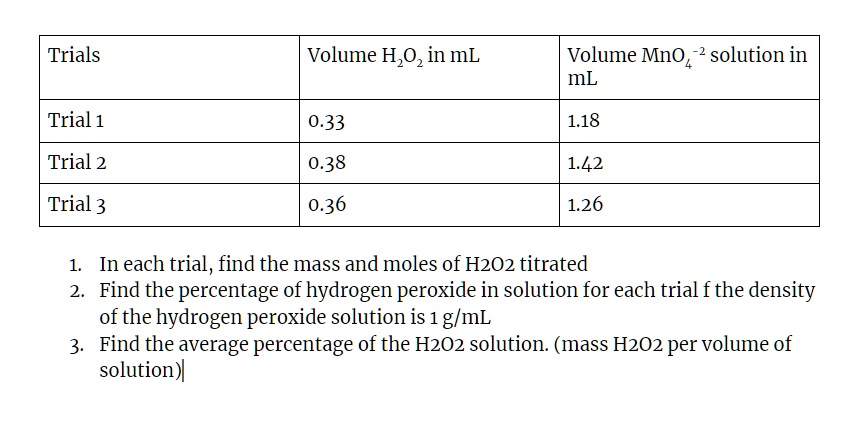
SOLVED: Text: Trials Volume H2O2 in mL Volume MnO4 solution in mL Trial 1 0.33 Trial 2 0.38 1.42 Trial 3 0.36 1.26 In each trial, find the mass and moles of
Calculate % (by mass) of a H2O2 solution which is 45.4 volume. - Sarthaks eConnect | Largest Online Education Community
The strength of 11.2 volume solution of H2O2 is : [Given that molar mass of H = 1g mol^–1 and O = 16g mol^–1] - Sarthaks eConnect | Largest Online Education Community