Assessment of the Quality of Reporting of Randomised Controlled Trials in Otorhinolaryngologic Literature – Adherence to the CONSORT Statement | PLOS ONE
![Example of abstract for report of pilot trial [21], shown alongside... | Download Scientific Diagram Example of abstract for report of pilot trial [21], shown alongside... | Download Scientific Diagram](https://www.researchgate.net/publication/309335377/figure/fig5/AS:419627092987905@1477058303192/Example-of-abstract-for-report-of-pilot-trial-21-shown-alongside-CONSORT-for-abstracts.png)
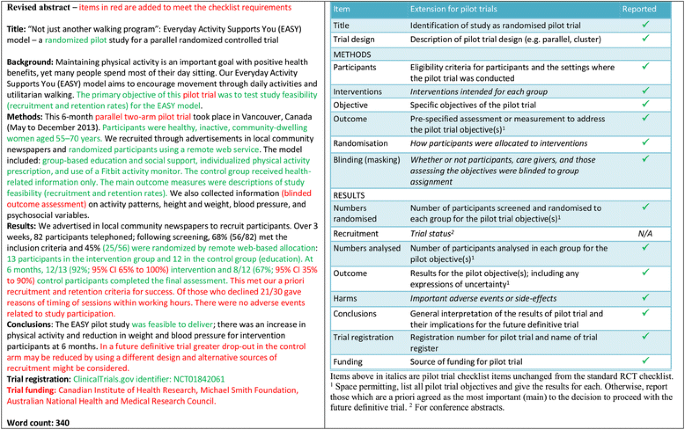
Example of abstract for report of pilot trial [21], shown alongside... | Download Scientific Diagram

PDF) CONSORT for Reporting Randomized Controlled Trials in Journal and Conference Abstracts: Explanation and Elaboration
CONSORT 2010 statement: extension to randomised pilot and feasibility trials | Pilot and Feasibility Studies | Full Text

CONSORT, QUOROM, and structured abstracts – new rules for authors, new tools for readers | European Journal of Anaesthesiology | Cambridge Core

Reporting quality for abstracts of randomised trials on child and adolescent depression prevention: a meta-epidemiological study on adherence to CONSORT for abstracts | BMJ Open

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension - The Lancet Digital Health

Did an introduction of CONSORT for abstracts guidelines improve reporting quality of randomised controlled trials' abstracts on Helicobacter pylori infection? Observational study | BMJ Open

Quality of reporting according to the CONSORT, STROBE and Timmer instrument at the American Burn Association (ABA) annual meetings 2000 and 2008 – topic of research paper in Clinical medicine. Download scholarly

Reporting of CONSORT for Abstracts items in articles with and without... | Download Scientific Diagram

Reported items in the modified 19-item CONSORT checklist for abstracts. | Download Scientific Diagram
CONSORT for reporting randomized controlled trials in journal and conference abstracts : Explanation and elaboration | QUT ePrints

Are we still missing the mark with randomised controlled trial abstracts? – The Publication Plan for everyone interested in medical writing, the development of medical publications, and publication planning
CONSORT for Reporting Randomized Controlled Trials in Journal and Conference Abstracts: Explanation and Elaboration | PLOS Medicine

Adherence to CONSORT for Abstracts – still room for improvement – The Publication Plan for everyone interested in medical writing, the development of medical publications, and publication planning









